Advertisements
Advertisements
प्रश्न
What is meant by the following term? Give an example of the reaction in the following case.
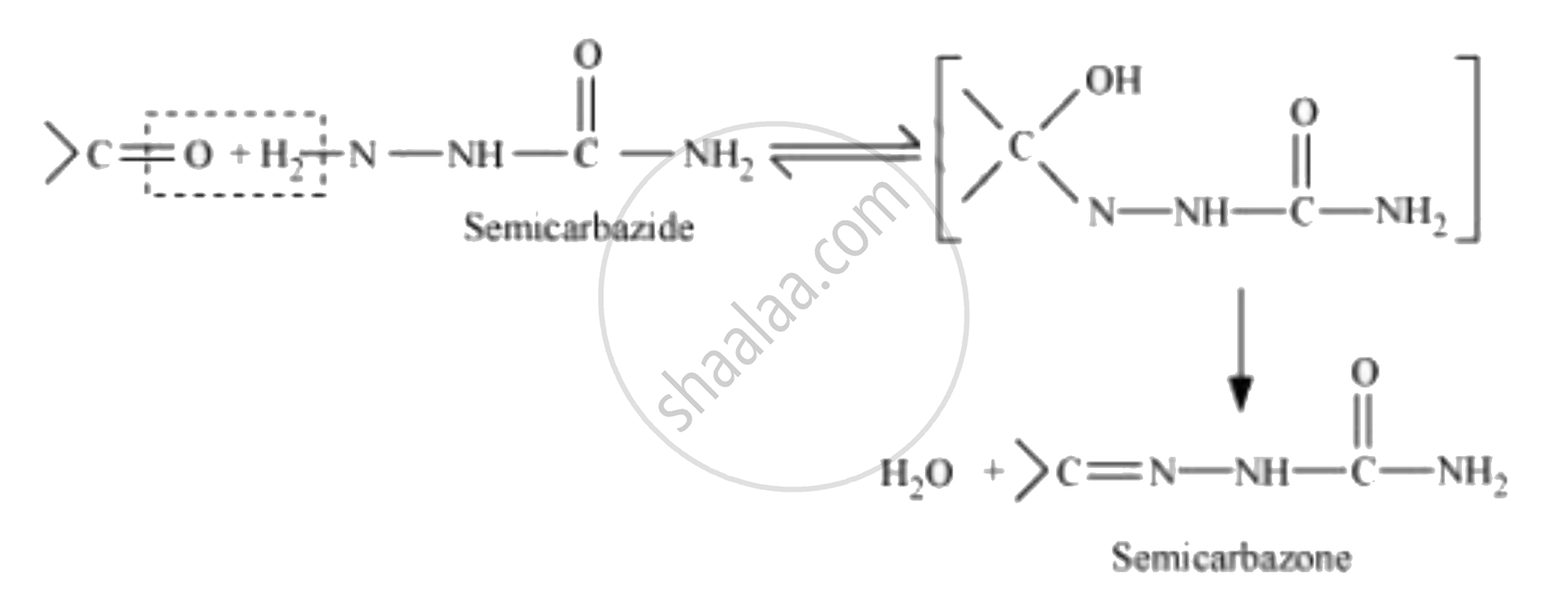
Semicarbazone
Advertisements
उत्तर
Semicarbazones are derivatives of aldehydes and ketones produced by the condensation reaction between a ketone or aldehyde and semicarbazide.
APPEARS IN
संबंधित प्रश्न
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Ethanal, Propanal, Propanone, Butanone.
Hint: Consider steric effect and electronic effect.
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Complete the synthesis by giving missing starting material, reagent or product.

Write balanced chemical equations for action of ammonia on - acetone
How will you convert sodium acetate to methane?
Give a simple chemical test to distinguish between 
Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
The most stable reagent for the conversion of R – CH2OH → RCHO is
What is the action of sodium hypoiodite on acetone?
What happens when propanone is treated with CH3MgBr and then hydrolysed?
The increasing order of the following compounds towards HCN addition is:
| (i) |  |
| (ii) |  |
| (iii) |  |
| (iv) |  |
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Draw structure of the following derivative.
The ethylene ketal of hexane-3-one
Why dissociation of HCN is suppressed by the addition of HCL?
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structures of the following derivatives.
Acetaldehydedimethylacetal
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Which of the following will not be formed when calcium formate is distilled with calcium acetate?
