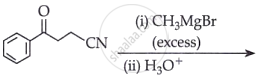Advertisements
Advertisements
प्रश्न
Write balanced chemical equations for action of ammonia on - acetone
Advertisements
उत्तर

APPEARS IN
संबंधित प्रश्न
Write the products formed when CH3CHO reacts with the following reagents : HCN
Predict the product of the following reaction:

Predict the product of the following reaction:
\[\begin{array}{cc}
\phantom{..............}\ce{O}\\
\phantom{..............}||\\
\ce{R - CH = CH - CHO + NH2 - C - NH - NH2 ->[H+]}\end{array}\]
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Excess ethanol and acid
Complete the synthesis by giving missing starting material, reagent or product.

What is the action of the following reagents on ethanoic acid?
1) `LiAlH_4"/"H_3O^+`
2) `PCl_3 , "heat"`
3) `P_2O_5, "heat"`
Acetone, Acetaldehyde, Benzaldehyde, Acetophenone – reactivity towards addition of HCN.
Write a test to differentiate between pentan-2-one and pentan-3-one.
Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
Grignard reagent on reaction with acetone forms.
The most stable reagent for the conversion of R – CH2OH → RCHO is
What is the action of sodium hypoiodite on acetone?
Which will undergo faster nucleophilic addition reaction?
Acetaldehyde or Propanone
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Draw the structure of the following derivative.
The ethylene ketal of hexane-3-one
Why dissociation of HCN is suppressed by the addition of HCL?
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
The product of the following reaction is
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{C2H5 - C - CH3 ->[H2/Ni][\Delta] \phantom{..}?}\end{array}\]
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structures of the following derivatives.
Acetaldehydedimethylacetal
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
The major product of the following reaction is: