Advertisements
Advertisements
Question
Write the products formed when CH3CHO reacts with the following reagents : H2N – OH
Advertisements
Solution 1
Ethanal reacts with hydroxylamine to form Ethanal oxime.
CH3CHO + H2NOH → CH3 - C = NOH + H2O
Ethanal oxime
Solution 2
Acetaldehyde (CH3CHO) reacts with Hydroxylamine (NH2OH) to give acetaldoxime as a product.

APPEARS IN
RELATED QUESTIONS
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Benzaldehyde, p-Tolualdehyde, p-Nitrobenzaldehyde, Acetophenone.
Hint: Consider steric effect and electronic effect.
Predict the product of the following reaction:

Which of the following is the correct representation for intermediate of nucleophilic addition reaction to the given carbonyl compound (A):
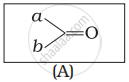
(i) 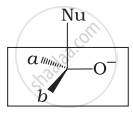
(ii) 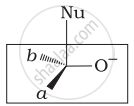
(iii) 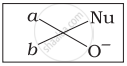
(iv) 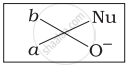
Alkenes 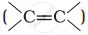 and carbonyl compounds
and carbonyl compounds 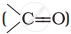 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
A Idol condensation will not be observed in
Which of the following is most reactive in nucleophilic addition reactions?
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
The product of the following reaction is
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{C2H5 - C - CH3 ->[H2/Ni][\Delta] \phantom{..}?}\end{array}\]
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Give an example of the reaction in the following case.
Oxime
