Advertisements
Advertisements
Question
Why pKa of F-CH2-COOH is lower than that of Cl−CH2−COOH?
Advertisements
Solution
Fluorine is more electronegative than chlorine. The presence of fluorine in a molecule will make it more acidic. Thus, the pKa of F−CH2−COOH is lower than that of Cl−CH2−COOH.
APPEARS IN
RELATED QUESTIONS
Which acid of the pair shown here would you expect to be stronger?
CH2FCO2H or CH2ClCO2H
Which acid of the pair shown here would you expect to be stronger?
CH2FCH2CH2CO2H or CH3CHFCH2CO2H
Which acid of the pair shown here would you expect to be stronger?
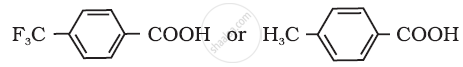
Arrange the following compounds in increasing order of their property as indicated:
CH3CH2CH(Br)COOH, CH3CH(Br)CH2COOH, (CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)
Arrange the following compounds in increasing order of their property as indicated:
Benzoic acid, 4-Nitrobenzoic acid, 3, 4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength)
Write the reactions involved Hell-Volhard Zelinsky reaction?
Account for the following:
Carboxylic acid is a stronger acid than phenol.
Complete the following reaction sequence.
\[\begin{array}{cc}
\ce{O}\phantom{...............................................}\\
||\phantom{...............................................}\\
\ce{CH3 - C - CH3 ->[(i) CH3MgBr][H2O] (A) ->[Na metal][Ether] (B) ->[CH3 - Br] (C)}
\end{array}\]
A mixture of benzaldehyde and formaldehyde on heating with 50% NaOH solution gives
When propionamide reacts with Br2 in the presence of alkali the product is ______.
