Advertisements
Advertisements
प्रश्न
Why pKa of F-CH2-COOH is lower than that of Cl−CH2−COOH?
Advertisements
उत्तर
Fluorine is more electronegative than chlorine. The presence of fluorine in a molecule will make it more acidic. Thus, the pKa of F−CH2−COOH is lower than that of Cl−CH2−COOH.
APPEARS IN
संबंधित प्रश्न
CH3CO2H or CH2FCO2H
Which acid of the pair shown here would you expect to be stronger?
CH2FCO2H or CH2ClCO2H
Which acid of the pair shown here would you expect to be stronger?
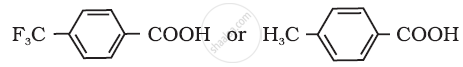
Account for the following:
Carboxylic acid is a stronger acid than phenol.
Complete the following reaction sequence.
\[\begin{array}{cc}
\ce{O}\phantom{...............................................}\\
||\phantom{...............................................}\\
\ce{CH3 - C - CH3 ->[(i) CH3MgBr][H2O] (A) ->[Na metal][Ether] (B) ->[CH3 - Br] (C)}
\end{array}\]
Acidity of BF3 can be explained on the basis of which of the following concepts?
A mixture of benzaldehyde and formaldehyde on heating with 50% NaOH solution gives
Na2CO3 cannot be used in place of (NH4)2CO3 for the precipitation of the V group because ______.
Formic acid and formaldehyde can be distinguished by treating with ______.
The correct order of decreasing acidity of the following aliphatic acids is:
