Advertisements
Advertisements
प्रश्न
What happens when ethanal is treated with excess ethanol and acid?
Advertisements
उत्तर
When ethanal reacts with excess ethanol and acid gives acetal.
\[\begin{array}{cc}
\phantom{.......}\ce{CH3}\phantom{.........................}\ce{CH3}\phantom{....}\ce{OC2H5}\\
\phantom{....}\backslash\phantom{..........................}\backslash\phantom{..}/\\
\ce{= O + \underset{(excess)}{C2H5OH} ->[H^⊕]}\\
\phantom{....}/\phantom{..........................}/\phantom{..}\backslash\\
\phantom{.........}\ce{H}\phantom{.........................}\ce{H}\phantom{.....}\ce{OC2H5}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
What is meant by the following term? Give an example of the reaction in the following case.
Oxime
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Write balanced chemical equations for action of ammonia on - acetone
What are amines?
Which one of the following gives only one monochloro derivative?
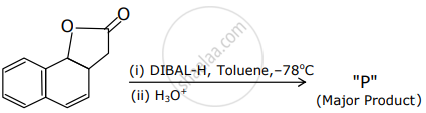
The product "P" in the above reaction is:
Which of the following is most reactive in nucleophilic addition reactions?
Draw structure of the following derivative.
The ethylene ketal of hexane-3-one
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
The product of the following reaction is
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{C2H5 - C - CH3 ->[H2/Ni][\Delta] \phantom{..}?}\end{array}\]
