Advertisements
Advertisements
प्रश्न
What is meant by the following term? Give an example of the reaction in the following case.
Oxime
Advertisements
उत्तर
Oximes are formed by reaction between aldehydes or ketones and hydroxylamine. The reaction is carried out in a mildly acidic condition.
\[\begin{array}{cc}
\ce{H3C}\phantom{...........................}\ce{H3C}\phantom{.....................}\\
\backslash\phantom{..............................}\backslash\phantom{................}\\
\ce{= O + \underset{Hydroxylarnine}{H2N-OH} ->[pH 3.5] \phantom{......}= NOH + H2O}\\
/\phantom{..............................}/\phantom{.................}\\
\ce{H}\phantom{............................}\ce{\underset{Oxime}{H}}\phantom{..................}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
What is meant by the following term? Give an example of the reaction in the following case.
Hemiacetal
What is meant by the following term? Give an example of the reaction in the following case.
Imine
What is meant by the following term? Give an example of the reaction in the following case.
2, 4-DNP-derivative
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Semicarbazide and weak acid
Explain the mechanism of alkaline hydrolysis of tert-butyl bromide with energy profile diagram.
Write balanced chemical equations for action of ammonia on - acetone
Give a simple chemical test to distinguish between 
Write the main product formed when propanal reacts with the following reagents:
H2N- NH2 followed by heating with KOH in ethylene glycol.
Alkenes 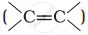 and carbonyl compounds
and carbonyl compounds 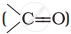 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
Acetaldehyde and acetone differ in their reaction with
What is the action of sodium hypoiodite on acetone?
The product of following reaction is
\[\ce{CH3 - CH = CH - CH2 - CHO ->[i) LiAlH4][ii) H3O+]}\] ______?
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Draw structures of the following derivatives.
Acetaldehydedimethylacetal
Draw structure of the following derivative:
Acetaldehydedimethylacetal
