Advertisements
Advertisements
प्रश्न
What is meant by the following term? Give an example of the reaction in the following case.
Oxime
Advertisements
उत्तर
Oximes are formed by reaction between aldehydes or ketones and hydroxylamine. The reaction is carried out in a mildly acidic condition.
\[\begin{array}{cc}
\ce{H3C}\phantom{...........................}\ce{H3C}\phantom{.....................}\\
\backslash\phantom{..............................}\backslash\phantom{................}\\
\ce{= O + \underset{Hydroxylarnine}{H2N-OH} ->[pH 3.5] \phantom{......}= NOH + H2O}\\
/\phantom{..............................}/\phantom{.................}\\
\ce{H}\phantom{............................}\ce{\underset{Oxime}{H}}\phantom{..................}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Acetaldehyde, when treated with which among the following reagents does NOT undergo addition reaction?
(A) Ammonia
(B) Hydroxylamine
(C) Ammoniacal silver nitrate
(D) Semicarbazide
Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction:
C6H5COCH3, CH3-CHO, CH3COCH3
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
H2N − NHCONH2/H+
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Ethanal, Propanal, Propanone, Butanone.
Hint: Consider steric effect and electronic effect.
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Benzaldehyde, p-Tolualdehyde, p-Nitrobenzaldehyde, Acetophenone.
Hint: Consider steric effect and electronic effect.
What is meant by the following term? Give an example of the reaction in the following case.
Cyanohydrin
What is meant by the following term? Give an example of the reaction in the following case.
2, 4-DNP-derivative
Explain the mechanism of alkaline hydrolysis of tert-butyl bromide with energy profile diagram.
What is the action of the following reagents on ethanoic acid?
1) `LiAlH_4"/"H_3O^+`
2) `PCl_3 , "heat"`
3) `P_2O_5, "heat"`
Write the structure of Phenylmethanamine.
Which of the following is the correct representation for intermediate of nucleophilic addition reaction to the given carbonyl compound (A):
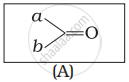
(i) 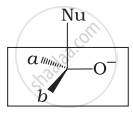
(ii) 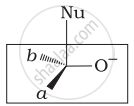
(iii) 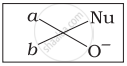
(iv) 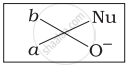
Which of the following has the most acidic hydrogen?
Grignard reagent on reaction with acetone forms.
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
Acetaldehydedimethylacetal
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
