Advertisements
Advertisements
Questions
Write the chemical equations to illustrate the following name reaction:
Cannizzaro’s reaction
Write the reactions involved in the following reaction:
Cannizzaro reaction
Advertisements
Solution
Aldehydes which do not have an α-hydrogen atom, undergo self-oxidation and reduction (disproportionation) reaction on treatment with a concentrated alkali.
\[\begin{array}{cc} \phantom{....}\ce{H}\phantom{........}\ce{H}\phantom{.........................}\ce{H}\phantom{...............}\ce{O}\\ \phantom{.....}\backslash\phantom{........}\backslash\phantom{.........................}|\phantom{...............}//\\ \ce{C = O + C = O + Conc{.} KOH ->[\Delta] H - C - OH + H - C}\\ \phantom{.....}/\phantom{........}/\phantom{.........................}|\phantom{...............}\backslash\\ \phantom{..........}\ce{\underset{Formaldehyde}{H\phantom{........}H}}\phantom{.......................}\ce{\underset{Methanol}{H}}\phantom{........}\ce{\underset{Potassium formate}{OK}} \end{array}\]
APPEARS IN
RELATED QUESTIONS
Write a note on the self oxidation-reduction reaction of an aldehyde with a suitable example.
An organic compound with the molecular formula C9H10O forms 2, 4-DNP derivative, reduces Tollens’ reagent and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1, 2-benzenedicarboxylic acid. Identify the compound.
Write the reactions involved in the following reactions: Clemmensen reduction
Complete the following reactions:

complete the following reaction:
\[\begin{array}{cc}
\phantom{...}\ce{CH3} \\
| \\
\phantom{.................}\ce{CH3-CH-COOH ->[(i) Br2/Red P4][(ii)H2O]}
\end{array}\]
Complete the following reaction:
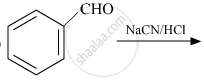
The products obtained in the Cannizzaro reaction are
The key step in cannizzaro reaction in the inter molecular shift qf
What happens when methanal undergoes cannizzaro reaction?
Which of the following does not give Cannizzaro reaction?
