Advertisements
Advertisements
प्रश्न
Write the chemical equations to illustrate the following name reaction:
Cannizzaro’s reaction
Write the reactions involved in the following reaction:
Cannizzaro reaction
Advertisements
उत्तर
Aldehydes which do not have an α-hydrogen atom, undergo self-oxidation and reduction (disproportionation) reaction on treatment with a concentrated alkali.
\[\begin{array}{cc} \phantom{....}\ce{H}\phantom{........}\ce{H}\phantom{.........................}\ce{H}\phantom{...............}\ce{O}\\ \phantom{.....}\backslash\phantom{........}\backslash\phantom{.........................}|\phantom{...............}//\\ \ce{C = O + C = O + Conc{.} KOH ->[\Delta] H - C - OH + H - C}\\ \phantom{.....}/\phantom{........}/\phantom{.........................}|\phantom{...............}\backslash\\ \phantom{..........}\ce{\underset{Formaldehyde}{H\phantom{........}H}}\phantom{.......................}\ce{\underset{Methanol}{H}}\phantom{........}\ce{\underset{Potassium formate}{OK}} \end{array}\]
APPEARS IN
संबंधित प्रश्न
Complete the following reactions:

Write the reactions involved in the following reactions: Clemmensen reduction
Write the equation involved in the following reaction:
Etard reaction
How will you convert acetone to acetone cyanohydrin?
complete the following reaction:
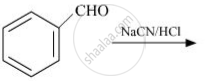
The products obtained in the Cannizzaro reaction are
Which of the following does not give Cannizzaro reaction?
Convert the following:
Benzene to m-nitrobenzaldehyde
Calcium acetate, on heating, gives:
Benzaldehyde can be prepared by the hydrolysis of:
