Advertisements
Advertisements
प्रश्न
Write the reactions involved in the following reactions: Clemmensen reduction
Advertisements
उत्तर
Clemmensen reduction: The carbonyl group of aldehydes and ketones is reduced to CH2 group on treatment with zinc-amalgam and concentrated hydrochloric acid is know as Clemmensen reduction.

APPEARS IN
संबंधित प्रश्न
Complete the following reactions:

Write the chemical equation for the reaction involved in Cannizzaro reaction.
Write the product(s) in the following reactions

Write the chemical equations to illustrate the following name reactions:
Aldol condensation
How will you convert acetone to acetone cyanohydrin?
complete the following reaction:
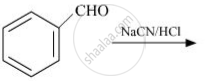
Complete the following reaction:
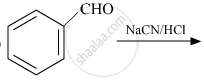
The key step in cannizzaro reaction in the inter molecular shift qf
In the Cannizzaro reaction given below:
\[\ce{2Ph-CHO ->[OH^-] Ph-CH2OH + PhC\overset{-}{O}_2}\]
the slowest step is:
Which of the following does not give Cannizzaro reaction?
