Advertisements
Advertisements
प्रश्न
Write a note on the self oxidation-reduction reaction of an aldehyde with a suitable example.
Advertisements
उत्तर
Cannizaro’s Reaction:-
(a) This is a characteristic reaction of those aldehydes which do not contain α -hydrogen atom.
(b) When such aldehydes are heated with concentrated alkali solutions like NaOH or KOH then simultaneous oxidation-reduction takes place.
(c) Out of two molecules of aldehyde one molecule, gets oxidised to form sodium or potassium salt of carboxylic acid and the second molecule is reduced to form corresponding primary alcohol.
(d) It is an auto oxidation-reduction reaction under the influence of base
(e) Formaldehyde and benzaldehyde undergo Cannizaro’s reaction as they do not contain α -hydrogen atom.
e.g.
\[\ce{\underset{\text{Formaldehyde(40-50%)}}{2H-CHO +NaOH}->[Redox][Reaction]\underset{\text{Sodium formate}}{HCOONa}+\underset{\text{Methanol}}{CH3}- OH}\]
\[\ce{\underset{\text{Benzaldehyde(50%)}}{2C6H5CHO +NaOH}->[Redox][Reaction]\underset{\text{Benzylalcohol}}{C6H5CH2OH} +\underset{\text{Sodiumbenzoate}}{C6H5COONa}}\]
(f) Acetaldehyde does not give this reaction since it contains α -Hydrogen atoms.
APPEARS IN
संबंधित प्रश्न
Complete the following reactions:

Write the chemical equation for the reaction involved in Cannizzaro reaction.
Write the chemical equations to illustrate the following name reaction:
Cannizzaro’s reaction
An organic compound with the molecular formula C9H10O forms 2, 4-DNP derivative, reduces Tollens’ reagent and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1, 2-benzenedicarboxylic acid. Identify the compound.
Write the reactions involved in the following reactions: Clemmensen reduction
Write the product(s) in the following reactions

Complete the following reactions:

Write the product formed when p-nitro chlorobenzene is heated with aqueous NaOH at 443K followed by acidification?
complete the following reaction:
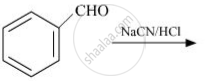
complete the following reaction:
\[\begin{array}{cc}
\phantom{...}\ce{CH3} \\
| \\
\phantom{.................}\ce{CH3-CH-COOH ->[(i) Br2/Red P4][(ii)H2O]}
\end{array}\]
The products obtained in the Cannizzaro reaction are
The key step in cannizzaro reaction in the inter molecular shift qf
Explain the following reaction:
Cannizzaro reaction
What happens when methanal undergoes cannizzaro reaction?
\[\begin{array}{cc}
\ce{D}\phantom{........................}\\
|\phantom{.........................}\\
\ce{2D - C = O + OH^- ->[Cannizzaro] X and Y}
\end{array}\]
(Y is alcohol, D is deuterium)
X and Y will have the structure:
In the Cannizzaro reaction given below:
\[\ce{2Ph-CHO ->[OH^-] Ph-CH2OH + PhC\overset{-}{O}_2}\]
the slowest step is:
Which of the following does not give Cannizzaro reaction?
Convert the following:
Benzene to m-nitrobenzaldehyde
Write the chemical reaction involved in Cannizzaro reaction of methanal.
Calcium acetate, on heating, gives:
