Advertisements
Advertisements
प्रश्न
Explain the following reaction:
Cannizzaro reaction
Explain the following reaction and write the chemical equation involved:
Cannizzaro reaction
Explain cannizzaro reaction with suitable example.
Advertisements
उत्तर
Aldehydes, which do not contain an α-hydrogen atom, undergo self-oxidation and reduction (disproportionation) reaction on heating with concentrated alkali (NaOH/KOH) to form an alcohol and a carboxylic acid salt.
\[\ce{\underset{Benzaldehyde}{2C6H5CHO} + NaOH -> \underset{Benzyl alcohol}{C6H5CH2OH} + \underset{Sodium benzoate}{C6H5COONa}}\]
APPEARS IN
संबंधित प्रश्न
Complete the following reactions:

Write a note on the self oxidation-reduction reaction of an aldehyde with a suitable example.
Write the chemical equations to illustrate the following name reaction:
Cannizzaro’s reaction
An organic compound with the molecular formula C9H10O forms 2, 4-DNP derivative, reduces Tollens’ reagent and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1, 2-benzenedicarboxylic acid. Identify the compound.
Describe the following:
Cannizzaro reaction
Write the reactions involved in the following reactions: Clemmensen reduction
Write the product(s) in the following reactions

Write the equation involved in the following reaction:
Etard reaction
Complete the following reactions:

Write the chemical equations to illustrate the following name reactions:
Aldol condensation
How will you convert acetone to acetone cyanohydrin?
Write the product formed when p-nitro chlorobenzene is heated with aqueous NaOH at 443K followed by acidification?
complete the following reaction:
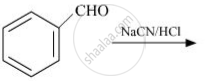
complete the following reaction:
\[\begin{array}{cc}
\phantom{...}\ce{CH3} \\
| \\
\phantom{.................}\ce{CH3-CH-COOH ->[(i) Br2/Red P4][(ii)H2O]}
\end{array}\]
Complete the following reaction:
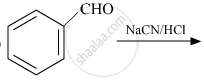
The products obtained in the Cannizzaro reaction are
What happens when methanal undergoes cannizzaro reaction?
\[\begin{array}{cc}
\ce{D}\phantom{........................}\\
|\phantom{.........................}\\
\ce{2D - C = O + OH^- ->[Cannizzaro] X and Y}
\end{array}\]
(Y is alcohol, D is deuterium)
X and Y will have the structure:
Which of the following does not give Cannizzaro reaction?
Convert the following:
Benzene to m-nitrobenzaldehyde
Calcium acetate, on heating, gives:
Which product is formed when the compound benzaldehyde is treated with concentrated aqueous KOH solution?
