Advertisements
Advertisements
Question
Distinguish between: CH3COOH and HCOOH
Advertisements
Solution
Formic acid gives the silver mirror test on treating with ammoniacal silver nitrate. Acetic acid does not give this test.
HCOOH + 2[Ag (NH3)2] NO3 →2Ag + CO2 + 2NH4NO3 + 2NH3
APPEARS IN
RELATED QUESTIONS
Distinguish between:
C6H5-COCH3 and C6H5-CHO
How will you convert ethanal into the following compound?
But-2-enoic acid
Give a simple chemical test to distinguish between the following pair of compounds:
Benzoic acid and Ethyl benzoate
Oxidation of ketones involves carbon-carbon bond cleavage. Name the products formed on oxidation of 2, 5-dimethylhexan-3-one.
Which of the following compounds gives a positive Tollen's test but negative Fehling's test?
Which of the following tests/reactions is given by aldehydes as well as ketones?
The major products formed in the following reaction sequence A and B are:

The correct set of products obtained in the following reactions:
- \[\ce{RCN ->[reduction]}\]
- \[\ce{RCN ->[(i) CH3MgBr][(ii) H2O]}\]
- \[\ce{RNC ->[hydrolysis]}\]
- \[\ce{RNH2 ->[HNO2]}\]
Choose the reaction which is not possible:
Match List-I with List-II:
| List-I (Reaction) |
List-II (Reagents/Condition) |
||
| A. | 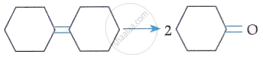 |
I. | 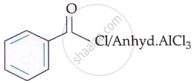 |
| B. | 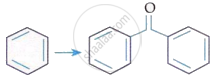 |
II. | CrO3 |
| C. | 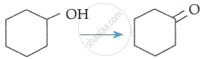 |
III. | KMnO4/KOH, Δ |
| D. |  |
IV. | (i) O3 (ii) Zn-H2O |
Choose the correct answer from the options given below:
