Advertisements
Advertisements
Question
Predict the products of the following reactions :

Advertisements
Solution
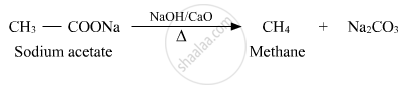
APPEARS IN
RELATED QUESTIONS
How will you bring about the following conversion in not more than two steps?
Benzoic acid to Benzaldehyde
Write the reactions involved Decarboxylation reaction
Give a reason for the following :
N-N bond is weaker than the P-P bond.
Which of the following is the strongest acid?
Which will undergo decarboxylation rapidly?
Maximum decarboxylation occurs in:
Identify product A in the following reaction.
\[\begin{array}{cc}
\ce{COOH}\phantom{.......}\\
/\phantom{...............}\\
\ce{CH2 ->[\Delta] CH3COOH + A}\\
\backslash\phantom{................}\\
\ce{COOH}\phantom{........}
\end{array}\]
Give the order of decarboxylation of the following acid:
| CH3COOH | CH2 = CH–CH2 – COOH |
| I | II |
| CH2(COOH)2 | 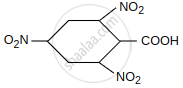 |
| III | IV |
The reactant (X) in the reaction
\[\begin{array}{cc}
\phantom{}\ce{\underset{}{(X)} ->[CH3COONa][(CH3CO)2O] Cinnamic acid, is}\\
\end{array}\]
In the following reaction, product P is:
\[\begin{array}{cc}
\phantom{....................}\ce{\underset{}{R - C - Cl} ->[H2][ Pd.BaSO4] \underset{}{P}}\\\phantom{}||\\\phantom{}\ce{O}\\
\end{array}\]
