Definitions [6]
Organic compounds containing carbon-oxygen double bond, i.e. \[\mathrm{>C=O}\] group, are known as carbonyl compounds.
The organic compounds in which the –OH group of a carboxylic acid is replaced by a halogen atom are called acyl halides.
The organic compounds in which the –OH group of a carboxylic acid is replaced by an –OR group are called esters.
The organic compounds formed by removal of one molecule of water from two molecules of carboxylic acid are called acid anhydrides.
The organic compounds in which the –OH group of a carboxylic acid is replaced by –NH₂ or substituted amino group are called amides.
The carbon–oxygen double bond (>C=O) functional group present in aldehydes, ketones and acids is called carbonyl group.
Formulae [4]
R–COX
R–COOR′
(R–CO)₂O
R–CONH₂
Chemica Equations [5]
\[2CH_3CHO\xrightarrow{dil.NaOH}CH_3CH(OH)CH_2CHO\]
On heating:
→CH3CH = CHCHO + H2O
\[RCOOH+R^{\prime}OH\xrightarrow{H^+}RCOOR^{\prime}+H_2O\]
\[RCHO\xrightarrow{NaBH_4}RCH_2OH\]
\[RCOOH\xrightarrow{LiAlH_4}RCH_2OH\]
\[RCOONa+NaOH\xrightarrow{CaO}RH+Na_2CO_3\]
Key Points
- Carbonyl group: The ≻C=O group (carbonyl carbon + carbonyl oxygen) — a key functional group in organic chemistry.
- Carbonyl compounds: Aldehydes and ketones, both containing ≻C=O as their functional group.
- Aldehydes: –CHO (formyl group); carbonyl C bonded to at least one H.
- Ketones: ≻C=O (ketonic carbonyl group); carbonyl C bonded to two alkyl/aryl groups (R=R′ or R≠R′).
- Carboxylic acids: –COOH (carboxyl group); –OH attached to ≻C=O makes them distinct from aldehydes/ketones.
In IUPAC system in aldehyde the suffix ‘e’ of alkane is replaced by ‘al’, e.g.,
CH₃—CH₂—CH=O; Propanal
\[ \underset{\text{2-methylpropanal}}{\mathrm{CH}_3 - \underset{\underset{\displaystyle \mathrm{CH}_3}{|}}{\mathrm{CH}} - \mathrm{CH} = \mathrm{O}} \]
In ketones, the suffix ‘e’ of alkane is replaced by ‘one’.
For example,
\[ \underset{\text{Butan-2-one}}{\mathrm{CH}_3 - \mathrm{CH}_2 - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{CH}_3} \quad \quad \underset{\text{Propanone (Acetone)}}{\mathrm{CH}_3\mathrm{COCH}_3} \]
- Carbon is sp² hybridised (trigonal planar, bond angle ≈ 120°).
- C = O bond consists of one σ bond and one π bond.
- Oxygen is more electronegative, so it pulls electron density towards itself →
C gets a partial positive charge (δ⁺) and O gets a partial negative charge (δ⁻). - This makes the carbonyl group polar.
- Hence, the carbon atom becomes electrophilic and is susceptible to nucleophilic attack.
Preparation of aliphatic aldehydes and ketones
By oxidation of alcohols:
\[\ \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{CH}-\mathrm{OH} \\ & / & \\ \mathrm{R}' & & \end{array} + [\mathrm{O}] \xrightarrow[\text{Or KMnO}_4]{\text{K}_2\text{Cr}_2\text{O}_7/\text{H}_2\text{SO}_4} \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{C}=\mathrm{O} \\ & / & \\ \mathrm{R}' & & \end{array} + \mathrm{H}_2\mathrm{O}\]
When,
- R' = H then 1° alcohol to aldehyde.
- R' = alkyl group then 2º alcohol to ketone.
By dehydrogenation of alcohols:
\[ \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{CH}-\mathrm{OH} \\ & / & \\ \mathrm{R}' & & \end{array} \xrightarrow[\text{573 K}]{\text{Cu}} \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{C}=\mathrm{O} \\ & / & \\ \mathrm{R}' & & \end{array} + \mathrm{H}_2 \]
When
- R' = H then 1° alcohol to aldehyde.
- R' = alkyl group the 2° alcohol to ketone.
By acid chloride:
\[ \mathrm{R} - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{Cl} + \mathrm{H}_2 \xrightarrow[\text{Rosenmund Reduction}]{\text{Pd}-\text{BaSO}_4} \underset{\text{Aldehyde}}{\mathrm{R} - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{H}} + \mathrm{HCl} \]
2RMgX + CdCl2 → R2Cd + 2MgXCl
\[\ 2\mathrm{R}' - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{Cl} + \mathrm{R}_2\mathrm{Cd} \longrightarrow 2\mathrm{R}' - \underset{\text{Ketone}}{\overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}}} - \mathrm{R} + \mathrm{CdCl}_2 \]
From nitriles and esters:
\[ \mathrm{R} - \mathrm{CN} \xrightarrow[\text{(ii) }\mathrm{H}_2\mathrm{O}]{\text{(i) }\mathrm{AlH}(i\text{Bu})_2} \underset{\text{Aldehyde}}{\mathrm{R} - \mathrm{CHO}} \]
\[ \mathrm{CH}_3(\mathrm{CH}_2)_9 - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{OC}_2\mathrm{H}_5 \xrightarrow[\text{(ii) }\mathrm{H}_2\mathrm{O}]{\text{(i) DIBAL-H}} \mathrm{CH}_3(\mathrm{CH}_2)_9 - \underset{\text{Aldehyde}}{\overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{H}} \]
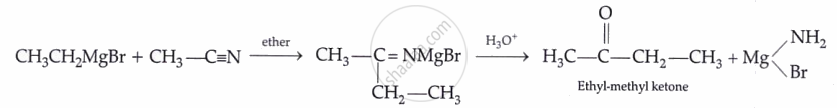 From hydrocarbons
From hydrocarbons
By ozonolysis:
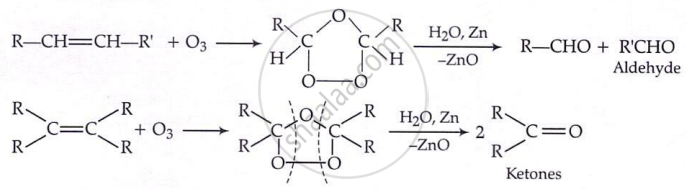
By hydration:
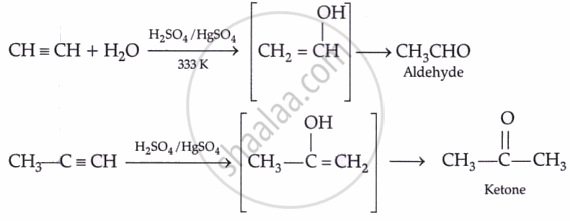
- Most aldehydes are liquids (except HCHO = gas); ketones of lower order are colourless liquids with a pleasant odour.
- Higher BP than corresponding hydrocarbons but lower than alcohols (no H-bonding between molecules, but dipole-dipole interactions).
- Lower members are soluble in water (H-bonding with water); higher members are insoluble (large alkyl groups).
Addition of HCN:
\[\text{HCN} + \text{OH}^- \rightleftharpoons :\text{CN}^- + \text{H}_2\text{O}\]
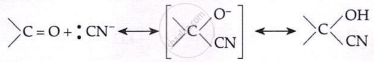
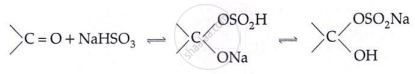
Addition of NaHSO3:
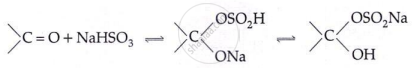
Addition of Grignard reagent:
Addition of alcohols:
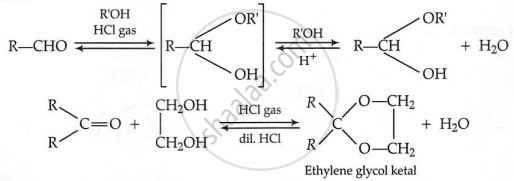
\[\text{R}-\text{CHO} + 2[\text{H}] \xrightarrow[\text{or BH}_3]{\text{LiAlH}_4} \text{RCH}_2\text{OH}\]
\[\begin{array}{r@{\;}c@{\;}l r@{\;}c@{\;}l} \ce{R} & & & \ce{R} & & \\ & \backslash & & & \backslash & \\ & & \ce{C=O + H2 ->[Ni or Pt]} & & & \ce{CH2OH} \\ & / & & & / & \\ \ce{R} & & & \ce{R} & & \end{array}\]
Clemmensen reduction:
\begin{array}{r@{\;}c@{\;}l c r@{\;}c@{\;}l} & & & & & & \\ & \backslash & & & & \backslash & \\ & & \ce{C=O} & \xrightarrow[\text{HCl}]{\text{Zn-Hg}} & & & \ce{CH2 + H2O} \\ & / & & & & / & \\ & & & & & & \end{array}
Wolff-Kishner reduction:
\begin{array}{r@{\;}c@{\;}l c r@{\;}c@{\;}l c r@{\;}c@{\;}l} & & & & & & & & & & \\ & \backslash & & & & \backslash & & & & \backslash & \\ & & \ce{C=O} & \xrightarrow[-\ce{H2O}]{\ce{NH2NH2}} & & & \ce{C=NNH2} & \xrightarrow{\text{KOH ethylene Glycol, }\Delta} & & & \ce{CH2 + N2} \\ & / & & & & / & & & & / & \\ & & & & & & & & & & \end{array}
\[\underset{\text{Aldehyde}}{\ce{R-CHO}} + \ce{[O]} \xrightarrow[\text{or }\ce{KMnO4/H2SO4}]{\ce{K2Cr2O7/H2SO4}} \underset{\text{Carboxylic acid}}{\ce{R-COOH}}\]
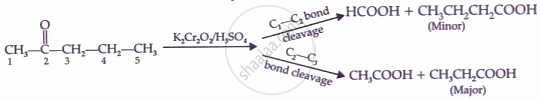
Aldol condensation:
\[\ce{CH3 - \overset{\displaystyle O}{\overset{||}{C}} - H + H - CH2 - \overset{\displaystyle O}{\overset{||}{C}} - H} \xrightarrow{\text{dil. NaOH}} \ce{H3C - \underset{\underset{\displaystyle OH}{|}}{CH} - \underset{\underset{\displaystyle H}{|}}{CH} - \overset{\displaystyle O}{\overset{||}{C}} - H} \\ \xrightarrow{\text{dil. }\ce{H2SO4}\Delta, \ce{-H2O}} \ce{CH3 - CH = CH - \overset{\displaystyle O}{\overset{||}{C}} - H}\]
Mechanism:
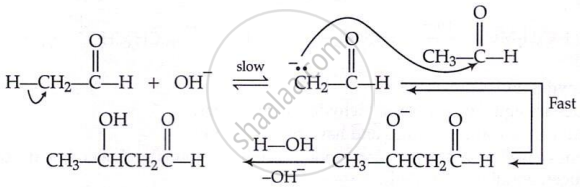
Cross aldol condensation:
\[\begin{array}{ccccccccccccc} & \ce{O} & & \ce{O} & & & & \ce{OH} & & \ce{O} & & & \\ & || & & || & & & & | & & || & & & \\ \ce{C6H5 -} & \ce{C} & \ce{+ HCH2 -} & \ce{C} & \ce{- H} & \overset{\text{dil. NaOH}}{\rightleftharpoons} & \ce{C6H5 -} & \ce{C} & \ce{- CH2 -} & \ce{C} & \ce{- H} & \xrightarrow[\Delta]{\ce{H2O}} & \ce{C6H5CH=CHCHO} \\ & | & & & & & & | & & & & & \text{Cinnamaldehyde} \\ & \ce{H} & & & & & & \ce{H} & & & & & \end{array}\]
Cannizzaro reaction: It is a self-oxidation reduction reaction.
\[\begin{array}{r@{\;}c@{\;}l} \ce{H} & & \\ & \backslash & \\ & & \ce{C=O} \\ & / & \\ \ce{H} & & \end{array} + \begin{array}{r@{\;}c@{\;}l} \ce{H} & & \\ & \backslash & \\ & & \ce{C=O} \\ & / & \\ \ce{H} & & \end{array} + \text{conc. KOH} \xrightarrow{\Delta} \ce{H - \underset{\underset{\displaystyle H}{|}}{\overset{\overset{\displaystyle H}{|}}{C}} - OH} + \ce{H - \overset{\displaystyle O}{\overset{||}{C}} - OK}\]
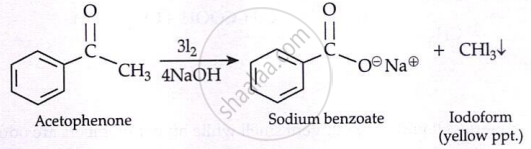
Haloform Reaction:
The reaction can be used to transform acetyl groups into carboxyl groups or to produce chloroform or iodoform. This reaction has been used in qualitative analysis to indicate the presence of a methyl ketone in which excess iodine is used to halogenate the compound. The product iodoform precipitates as a yellow-coloured substance and has a characteristic odour.
\[\underset{\substack{\text{Acetone} \\ \text{Iodoform Test}}}{\ce{H3C - \overset{\displaystyle O}{\overset{||}{C}} - CH3}} \xrightarrow[4\ce{NaOH}]{3\ce{Cl2}} \underset{\text{Sodium acetate}}{\ce{H3C - \overset{\displaystyle O}{\overset{||}{C}} - O^\ominus Na^\oplus}} + \underset{\text{Chloroform}}{\ce{CHCl3}}\]
- Formaldehyde: Used in making Bakelite (phenol-formaldehyde resin), as a preservative (formalin = 40% HCHO).
- Acetaldehyde: Used in the preparation of acetic acid and ethanol.
- Acetone: Solvent (nail polish remover), used in the manufacture of chloroform.
- Benzaldehyde: Used in perfumes and dyes.
- Special preparations: Benzoic acid from cumene (KMnO₄/KOH, Δ → H₃O⁺); adipic acid from cyclohexene (KMnO₄/dil. H₂SO₄, Δ).
- Aldehyde preparation: From 1° alcohol (K₂Cr₂O₇/H₂SO₄ or Cu/573 K), alkene (ozonolysis), alkyne (dil. H₂SO₄/HgSO₄), acid chloride (Rosenmund), nitrile (Stephen/DIBAL-H); aromatic via Etard, CrO₃/(CH₃CO)₂O, Cl₂/hν, Gatterman–Koch.
- Aldehyde reactions: HCN → cyanohydrin; NaHSO₃ → bisulphite adduct; R′OH → acetal; RMgBr → 2° alcohol; NH₂OH → aldoxime; NH₂NH₂ → hydrazone; K₂Cr₂O₇ → COOH; Clemmensen/Wolf–Kishner → alkane.
- Carboxylic acid preparation: From nitriles (hydrolysis), acyl chloride/anhydride/ester + H₂O, CO₂ + RMgX (dry ether, H₃O⁺), alkylbenzene (KMnO₄–KOH).
- Carboxylic acid reactions: PCl₅/SOCl₂ → RCOCl; NH₃ → amide; P₂O₅ → anhydride; NaOH + CaO (Δ) → alkane; LiAlH₄ → 1° alcohol; ROH/conc. H₂SO₄ → ester.
- Solubility: Decreases with an increase in the size of the hydrocarbon part.
- Miscibility: Lower carboxylic acids (up to 4 C atoms) are miscible with water due to H-bonding.
- Boiling point: Carboxylic acids have higher B.P. than ketones, aldehydes, and alcohols of comparable molecular mass due to intermolecular H-bonding.
- Order of B.P. (carboxylic acids & aldehydes): Valeric > Butyric > Propionic > Acetic > Formic acid; Hexanal > Pentanal > Butanal > Propanal.
- Order of B.P. (ketones): Hexan-2-one > Pentan-2-one > Butan-2-one > Propanone.
- Methanoic acid (Formic acid): Leather tanning, dyeing and finishing in textiles.
- Ethanoic acid (Acetic acid): Manufacturing of rayon and plastic; used as vinegar in cooking.
- Benzoic acid: Used as a food preservative and in perfumery.
- Salicylic acid: Used in the preparation of Aspirin (analgesic/antipyretic), Salol, and Oil of Wintergreen (methyl salicylate).
- Aspirin = acetylsalicylic acid; anti-pyretic and pain killer.
- Methyl salicylate = Oil of wintergreen (from methanol + salicylic acid).
Statement:
Carbonyl compounds undergo reduction to alcohols or hydrocarbons depending on reagents used.
Reduction to Alcohols
Reagents:
- NaBH₄
- LiAlH₄
- Catalytic hydrogenation (H₂/Ni)
Reactions:
Aldehyde → Primary alcohol
R–CHO → R–CH₂OH
Ketone → Secondary alcohol
R–CO–R′ → R–CHOH–R′
Reduction to Hydrocarbons
1. Clemmensen Reduction:
Zn(Hg)/HCl
R–CO–R′ → R–CH₂–R′
2. Wolff–Kishner Reduction:
NH₂NH₂/KOH
R–CO–R′ → R–CH₂–R′
Key Points:
- Carbonyl group converted to CH₂ group.
- Choice of reagent depends on acidic/basic conditions.
Conclusion:
Carbonyl compounds can be selectively reduced to alcohols or completely reduced to hydrocarbons.
Statement:
Aldehydes are more reactive than ketones towards nucleophilic addition reactions.
Reasons:
1. Steric Effect
- Aldehydes have: One alkyl group + one hydrogen.
- Ketones have: Two alkyl groups.
- More alkyl groups → more steric hindrance → less reactivity.
2. Electronic Effect (+I Effect)
- Alkyl groups show +I effect.
- They donate electron density to carbonyl carbon.
- This reduces partial positive charge on carbon.
Ketones (two alkyl groups) are less electrophilic.
Order of Reactivity:
Formaldehyde > Other aldehydes > Ketones
Example:
HCHO > CH₃CHO > CH₃COCH₃
Conclusion:
Due to lower steric hindrance and higher electrophilicity, aldehydes react faster than ketones.
Statement:
The carbonyl carbon atom is sp² hybridised, forms a trigonal planar structure, and possesses a polar C=O bond.
Explanation:
1. Hybridisation:
- Carbonyl carbon is sp² hybridised.
- It forms:
- Three σ (sigma) bonds.
- One π (pi) bond with oxygen.
- The π-bond is formed by sideways overlap of p-orbitals.
2. Geometry:
- The carbonyl carbon and three attached atoms lie in the same plane.
- Bond angle ≈ 120°.
- Geometry is trigonal planar.
3. Polarity:
- Oxygen is more electronegative than carbon.
- Hence, electron density shifts towards oxygen.
- Carbon acquires partial positive charge (δ⁺).
- Oxygen acquires partial negative charge (δ⁻).
4. Resonance:
Two contributing structures:
R–C=O ↔ R–C⁺–O⁻
This explains:
- High dipole moment.
- Electrophilic nature of carbonyl carbon.
Conclusion:
The planar structure, polarity and resonance make the carbonyl carbon highly reactive towards nucleophiles.
Important Questions [123]
- Give Simple Chemical Tests to Distinguish Between the Following Pairs of Compounds: Butanal and Butan-2-one
- Write the Structure of 2-methylbutanal.
- Using IUPAC norms, write the formula for the following: Pentaamminenitrito-N-Cobalt (III)
- Write the IUPAC name of the following complex: [Pt(NH3)6]Cl4
- Write IUPAC name of the following compound:
- Complete the following: CHA3CN→2⋅HA2O1⋅AlH(i−Bu)A2A′A22′AA′→HA+HA2N−OHA′A22′BA′
- Write the IUPAC name of the following complex: K2[PdCl4]
- Give the IUPAC name of the following compound. CH3 − CH = CH − CHO
- Write the Structure of 3-methyl Butanal
- Using IUPAC norms write the formula for the following: Tetrahydroxidozincate (II)
- Write the Structures of a and B in the Following Reactions
- Write the Iupac Name of the Following Compound:
- Complete synthesis by giving missing starting material, reagent or product. →[(i) O3][(ii) Zn-H2O] 2 O
- Complete the Following Reaction:
- Arrange the following in the increasing order of their boiling points: CH3CHO, CH3COOH, CH3CH2OH
- Give a chemical test to distinguish between ethanal acid and ethanoic acid.
- Why is the boiling point of aldehydes and ketones lower than that of corresponding carboxylic acids?
- Predict the Products of the Following Reactions
- Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction: C6H5COCH3, CH3-CHO, CH3COCH3
- Draw the structure of the semicarbazone of ethanal.
- Write the Structures of the Main Products When Acetone (CH3 − CO − CH3) Reacts with the Following Reagent
- Write the products formed when CH3CHO reacts with the following reagents : HCN
- Write the products formed when CH3CHO reacts with the following reagents : H2N – OH
- Give a Simple Chemical Test to Distinguish Between
- Write the Main Product Formed When Propanal Reacts with the Following Reagents:
- Write the main product formed when propanal reacts with the following reagents: H2N- NH2 followed by heating with KOH in ethylene glycol.
- Acetone, Acetaldehyde, Benzaldehyde, Acetophenone – Reactivity Towards Addition of Hcn.
- Arrange the following in the increasing order of their property indicated: Ethanal, Propanone, Propanal, Butanone (reactivity towards nucleophilic addition)
- What happens when propanone is treated with CH3MgBr and then hydrolysed?
- What happens when ethanal is treated with excess ethanol and acid?
- Which will undergo faster nucleophilic addition reaction? Acetaldehyde or Propanone
- Which of the following is most reactive in nucleophilic addition reactions?
- The carbon-oxygen double bond is polarised in aldehydes and ketones due to higher electronegativity of oxygen relative to carbon.
- Aldehydes and ketones react with hydroxylamine to form ______.
- Predict the Products of the Following Reactions : C H 3 C H 2 O H → C U / 573 K ?
- Write the chemical reaction involved in Wolff-Kishner reduction.
- Etrh
- Predict the Products of the Following Reactions
- Explain the following reaction: Wolff-Kishner reduction
- Write Chemical Equations of the Following Reaction : Acetophenone is Treated with ("Zn"("Hg"))/"Conc.Hcl"^-
- Predict the Products of the Following Reactions : C H 3 − C H = C H 2 → I ) B 2 H 6 I I ) 3 H 2 O 2 / O H − ?
- How Will You Bring About the Following Conversions? Propanone to Propane
- Write the main product in the following reaction: CHA3CHA2CHO→Zn(Hg)/Conc⋅HCl
- Explain the following reactions: Clemmensen reaction
- Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent
- Out of CH3CH2 – CO – CH2 – CH3 and CH3CH2 – CH2 – CO – CH3, which gives iodoform test?
- A and B are two functional isomers of compound C3H6O.On heating with NaOH and I2, isomer B forms yellow precipitate of iodoform whereas isomer A does not form any precipitate. Write the formulae of A and B.
- What is the composition of Fehling's reagent?
- An organic compound 'A' with molecular formula C5H8O2 is reduced to n-pentane with hydrazine followed by heating with NaOH and glycol. 'A' forms a dioxime with hydroxylamine
- Give a simple chemical test to distinguish between the following pair of compounds: Benzaldehyde and Acetophenone
- An organic compound 'A' with the molecular formula C4H8O2 undergoes acid hydrolysis to form two compounds 'B' and 'C'.
- Write chemical test to distinguish between the following compounds: Phenol and Benzoic acid
- C 6 H 5 − O H → B R 2 ( a Q ) ?
- Distinguish Between: C6H5-COCH3 and C6H5-CHO
- Give a simple chemical test to distinguish between the following pair of compounds : CH3CH2CHO and CH3CH2COCH3
- Distinguish Between: CH3COOH and HCOOH
- Give a simple chemical test to distinguish between the following pair of compounds: Ethanal and Propanal
- Give a simple chemical test to distinguish between the following pair of compounds: Propanal and Propanone
- The reagent that can be used to distinguish acetophenone and benzophenone is ______.
- Predict the Products of the Following Reactions
- How will you convert ethanal into the following compound? But-2-enal
- Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?
- Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
- Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
- How will you bring about the following conversion in not more than two steps? Ethanol to 3-Hydroxybutanal
- Write Chemical Equations of the Following Reaction : Benzoyl Chloride is Hydrogenated in the Presence of "Pd"/("Baso"4^-)
- Write a Chemical Equation for the Following Reaction : Propanone is Treated with Dilute Ba( Oh)2-.
- Give Reasons Acetylation of Aniline Reduces Its Activation Effect.
- A Compound 'A' of Molecular Formula C2H3OCl Undergoes a Series of Reactions as Shown Below. Write the Structures of A, B, C and D in the Following Reactions
- Which of the following does not give aldol condensation reaction?
- Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound. Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
- Predict the reagent for carrying out the following transformations: Ethanal to 3-hydroxy butanal
- Convert the following: Acetaldehyde to But-2-enal
- Why is Alpha (α) Hydrogen of Carbonyl Compounds Acidic in Nature?
- Write the Product Formed When P-nitro Chlorobenzene is Heated with Aqueous Naoh at 443k Followed by Acidification?
- Write the chemical equations to illustrate the following name reaction: Cannizzaro’s reaction
- Write the Reactions Involved in the Following Reactions: Clemmensen Reduction
- Write the Product(S) in the Following Reactions -1
- Write the equation involved in the following reaction: Etard reaction
- Complete the Following Reactions
- Complete the Following Reactions
- Write the Chemical Equations to Illustrate the Following Name Reactions: Aldol Condensation
- Which of the following does not give Cannizzaro reaction?
- Complete the Following Reaction:
- Complete the Following Reaction:
- Complete the Following Reaction:
- Explain the following reaction: Cannizzaro reaction
- Complete the Following Reactions
- What happens when methanal undergoes cannizzaro reaction?
- Write the Chemical Equation for the Reaction Involved in Cannizzaro Reaction.
- Write the Structure of 2-hydroxybenzoic Acid.
- How do you convert toluene to benzoic acid?
- Write the Equations Involved in the Following Reactions : Wolff-kishner Reduction
- Write the Equations Involved in the Following Reactions : Stephen Reaction
- How do you convert the following: Acetic acid to Methane
- How Do You Convert the Following : Ethyne to Ethanal
- Account for the Following: Aromatic Carboxylic Acids Do Not Undergo Friedel-crafts Reaction.
- Why Carboxylic Acid Does Not Give Reactions of Carbonyl Group?
- Write the structures of A and B in the following reactions
- An organic compound (A) (molecular formula C8H16O2) was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) and an alcohol (C). Oxidation of (C) with chromic acid produced (B).
- Predict the Products of the Following Reactions
- Name the Reagents Used in the Following Reactions
- Name the Reagents Used in the Following Reactions:
- Why Are Alkyl Halides Insoluble in Water?
- Account for the Following: Pka Value of 4-nitrobenzoic Acid is Lower than that of Benzoic Acid.
- Arrange the Following Compounds in Increasing Order of Their Property as Indicated:
- What Happens When Salicylic Acid is Treated with (Ch3co)2 O/H+?
- Account for the Following: Carboxylic Acid is a Stronger Acid than Phenol.
- Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Why?
- Why Pka of F-CH2-COOH is Lower than that of Cl−CH2−COOH?
- Write the Reactions Involved Hell-volhard Zelinsky Reaction?
- Distinguish Between the Following : Benzoic Acid and Methyl Benzoate
- Draw the Structures of Major Monohalo Products in Each of the Following Reactions
- Name the reagents used in the following reactions : CH3-COOH → ClCH2-COOH
- Predict the Products of the Following Reactions
- Give a Reason for the Following : N-n Bond is Weaker than the P-p Bond.
- Write the Reactions Involved Decarboxylation Reaction
- Predict the Products of the Following Reactions
- How will you bring about the following conversion in not more than two steps? Benzoic acid to Benzaldehyde
- Assertion (A): Acetic acid but not formic acid, can be halogenated in the presence of red P and Cl2. Reason (R): Acetic acid is a weaker acid than formic acid.
- Account for the following : Cl – CH2COOH is a stronger acid than CH3COOH.
- Convert the following: Propan-1-ol to 2-Bromopropanoic acid
- Write the main product in the following reaction: COONa + NaOH ->[CaO][Delta]
Concepts [21]
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Nomenclature of Aldehydes and Ketones
- Structure of Carbonyl Group
- Preparation of Aldehydes and Ketones
- Physical Properties of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
- Chemical Reactions of Aldehydes and Ketones - Reduction
- Chemical Reactions of Aldehydes and Ketones - Oxidation
- Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
- Chemical Reactions of Aldehydes and Ketones - Other Reactions
- Uses of Aldehydes and Ketones
- Nomenclature of Carboxylic Acids
- Structure of the Carboxyl group
- Methods of Preparation of Carboxylic Acids
- Physical Properties of Carboxylic Acids
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of C-OH Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving –COOH Group
- Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
- Uses of Carboxylic Acids
- Overview: Aldehydes, Ketones and Carboxylic Acids
