Advertisements
Advertisements
Question
Assertion (A): Acetic acid but not formic acid, can be halogenated in the presence of red P and Cl2.
Reason (R): Acetic acid is a weaker acid than formic acid.
Options
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Advertisements
Solution
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Explanation:
Acetic acid is halogenated in the presence of red P and Cl2, but formic acid is not because it lacks an α-hydrogen atom. In comparison to formic acid, proton loss becomes more difficult. As a result, acetic acid is a weaker acid than formic acid.
APPEARS IN
RELATED QUESTIONS
Account for the following : Cl – CH2COOH is a stronger acid than CH3COOH.
Write chemical equation for the following :
Hell - Vohlard - Zelinsky reaction
Convert the following:
Propan-1-ol to 2-Bromopropanoic acid
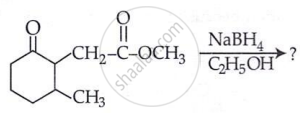
The product formed in the following chemical reaction is
Propionic acid with Br2/P yields a dibromo product. Its structure would be:
