Advertisements
Advertisements
Question
A and B are two functional isomers of compound C3H6O.On heating with NaOH and I2, isomer B forms yellow precipitate of iodoform whereas isomer A does not form any precipitate. Write the formulae of A and B.
Advertisements
Solution

APPEARS IN
RELATED QUESTIONS
Distinguish between: CH3COOH and HCOOH
Predict the products of the following reactions :

Complete the synthesis by giving missing starting material, reagent or product.

Solvent used for dewaxing of petroleum products are
Ammonical silver nitrate solution is called
Write chemical test to distinguish between the following compounds:
Phenol and Benzoic acid
An organic compound neither reacts with neutral ferric chloride solution nor with Fehling solution. It however, reacts with Grignard reagent and gives positive iodoform test. The compound is:
Choose the reaction which is not possible:
Benzaldehyde is obtained from Rosenmund’s reduction of:
Match List-I with List-II:
| List-I (Reaction) |
List-II (Reagents/Condition) |
||
| A. | 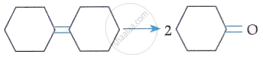 |
I. | 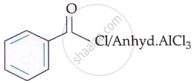 |
| B. | 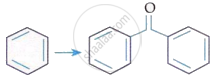 |
II. | CrO3 |
| C. | 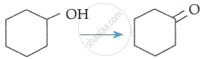 |
III. | KMnO4/KOH, Δ |
| D. | 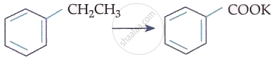 |
IV. | (i) O3 (ii) Zn-H2O |
Choose the correct answer from the options given below:
