Advertisements
Advertisements
Question
Which of the following does not give aldol condensation reaction?
Options
CH3 – CHO
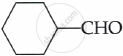
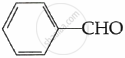
CH3COCH3
Advertisements
Solution

Explanation:
Aldehydes and ketones with at least one α-hydrogen undergo a reaction in the presence of dilute alkali as a catalyst to create β-hydroxy aldehydes (aldol) or β-hydroxy ketones (ketol), respectively. This is known as an Aldol reaction.
Due to the lack of α-hydrogen, benzaldehyde does not undergo aldol reaction.
APPEARS IN
RELATED QUESTIONS
Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
How will you bring about the following conversion in not more than two steps?
Benzaldehyde to 3-Phenylpropan-1-ol
Write chemical equations of the following reaction :
Benzoyl chloride is hydrogenated in the presence of `"Pd"/(BaSO_4)`
Write a chemical equation for the following reaction:
Propanone is treated with dilute Ba( OH)2.
Compounds A and C in the following reaction are:
\[\ce{CH3CHO ->[(i) CH3MgBr][(ii) H2O] (A) ->[H2SO4, Δ] (B) ->[Hydroboration oxidation] (C)}\]
Why is there a large difference in the boiling points of butanal and butan-1-ol?
Cross aldol condensation occurs between
Which of the following gives aldol con~ensation reaction?
Convert the following:
Acetaldehyde to But-2-enal
Explain Aldol condensation of ethanal.
