Advertisements
Advertisements
Question
Give a simple chemical test to distinguish between the following pair of compounds :
CH3CH2CHO and CH3CH2COCH3
Advertisements
Solution
2-Butanone has a methyl group attached to carbonyl carbon unlike propanal. Hence, it can give iodofom test.

RELATED QUESTIONS
A and B are two functional isomers of compound C3H6O.On heating with NaOH and I2, isomer B forms yellow precipitate of iodoform whereas isomer A does not form any precipitate. Write the formulae of A and B.
Propanal and Propanone
Alkenes decolourise bromine water in presence of CCl4 due to formation of ______.
Which of the following compounds will give butanone on oxidation with alkaline \[\ce{KMnO4}\] solution?
Fehilng's test is positive for
What is the composition of Fehling's reagent?

Which among the above compound/s does/do not form Silver mirror when treated with Tollen's reagent?
The major products formed in the following reaction sequence A and B are:

The reagent that can be used to distinguish acetophenone and benzophenone is ______.
Match List-I with List-II:
| List-I (Reaction) |
List-II (Reagents/Condition) |
||
| A. | 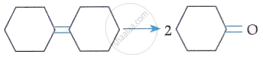 |
I. | 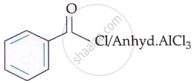 |
| B. | 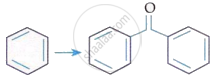 |
II. | CrO3 |
| C. | 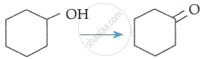 |
III. | KMnO4/KOH, Δ |
| D. |  |
IV. | (i) O3 (ii) Zn-H2O |
Choose the correct answer from the options given below:
