Advertisements
Advertisements
Question
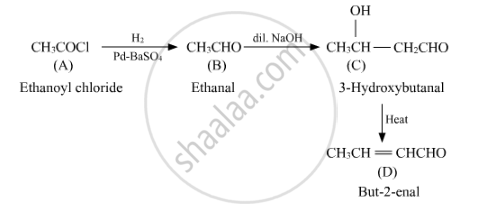
A compound 'A' of molecular formula C2H3OCl undergoes a series of reactions as shown below. Write the structures of A, B, C and D in the following reactions :

Advertisements
Solution
Using the given molecular formula, compound A is Ethanoyl Chloride CH3COCl, which undergoes reaction with poisoned palladium.
On carrying hydrogenation of A in the presence of poisoned palladium, we get an aldehyde. Hence, B can be Ethanal, CH3CHO.
An aldehyde, on treating with dilute alkali, undergoes aldol condensation reaction. Hence, C can be CH3CH(OH)CH2CHO.
On heating an aldol product, it loses water to produce a double bond and we get CH3CH=CHCHO.
Hence, we have
RELATED QUESTIONS
How will you convert ethanal into the following compound?
But-2-enal
How will you bring about the following conversion in not more than two steps?
Ethanol to 3-Hydroxybutanal
Write chemical equations of the following reaction :
Benzoyl chloride is hydrogenated in the presence of `"Pd"/(BaSO_4)`
Write a chemical equation for the following reaction:
Propanone is treated with dilute Ba( OH)2.
Which product is formed when the compound  is treated with concentrated aqueous \[\ce{KOH}\] solution?
is treated with concentrated aqueous \[\ce{KOH}\] solution?
Why is there a large difference in the boiling points of butanal and butan-1-ol?
When liquid ‘A’ is treated with a freshly prepared ammoniacal silver nitrate solution, it gives bright silver mirror. The liquid forms a white crystalline solid on treatment with sodium hydrogensulphite. Liquid ‘B’ also forms a white crystalline solid with sodium hydrogensulphite but it does not give test with ammoniacal silver nitrate. Which of the two liquids is aldehyde? Write the chemical equations of these reactions also.
Give reasons to support the answer:
Presence of Alpha hydrogen in aldehydes and ketones is essential for aldol condensation.
\[\ce{CH3-CH2-CHO ->[dil][alkali] Product}\]
The product in the above reaction is:
What is aldol condensation? Explain it with suitable examples.
