Advertisements
Advertisements
Question
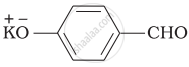
Which product is formed when the compound  is treated with concentrated aqueous \[\ce{KOH}\] solution?
is treated with concentrated aqueous \[\ce{KOH}\] solution?
Options
Advertisements
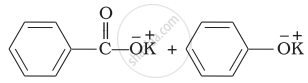
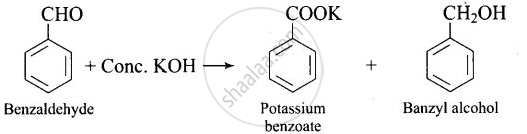
Solution

Explanation:
Benzaldehyde is having no a hydrogen. So, on reaction with aqueous KOH solution, it undergoes Cannizzaro’s reaction. One molecule of aldehyde is reduced and other is
APPEARS IN
RELATED QUESTIONS
How will you convert ethanal into the following compound?
Butane-1, 3-diol
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile.
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
What is substituted imine called?
Which of the following compounds do not undergo aldol condensation?
(i) \[\ce{CH3 - CHO}\]
(ii) 
(iii) \[\begin{array}{cc}
\phantom{}\ce{O}\\
\phantom{}||\\
\ce{CH3 - C - CH3}
\end{array}\]
(iv) \[\begin{array}{cc}
\phantom{}\ce{CH3}\\
|\phantom{...}\\
\ce{CH3 - C - CHO}\phantom{..}\\
|\phantom{...}\\
\phantom{}\ce{CH3}\\
\end{array}\]
Which of the following conversions can be carried out by Clemmensen Reduction?
(i) Benzaldehyde into benzyl alcohol
(ii) Cyclohexanone into cyclohexane
(iii) Benzoyl chloride into benzaldehyde
(iv) Benzophenone into diphenyl methane
Why are carboxylic acids more acidic than alcohols or phenols although all of them have hydrogen atom attached to a oxygen atom \[\ce{(-O-H)}\]?
Assertion: Aromatic aldehydes and formaldehyde undergo Cannizaro reaction.
Reason: Aromatic aldehydes are almost as reactive as formaldehyde.
Give reasons to support the answer:
Presence of Alpha hydrogen in aldehydes and ketones is essential for aldol condensation.
Which of the following gives aldol con~ensation reaction?
Convert the following:
Acetaldehyde to But-2-enal
Predict the reagent for carrying out the following transformations:
Ethanal to 3-hydroxy butanal
\[\ce{CH3-CH2-CHO ->[dil][alkali] Product}\]
The product in the above reaction is:
The major product of the following reaction is:

Explain Aldol condensation of ethanal.
Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?
Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound.
Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
What is aldol condensation? Explain it with suitable examples.