Advertisements
Advertisements
प्रश्न
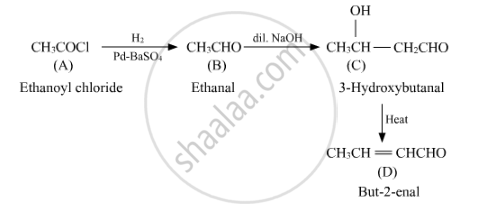
A compound 'A' of molecular formula C2H3OCl undergoes a series of reactions as shown below. Write the structures of A, B, C and D in the following reactions :

Advertisements
उत्तर
Using the given molecular formula, compound A is Ethanoyl Chloride CH3COCl, which undergoes reaction with poisoned palladium.
On carrying hydrogenation of A in the presence of poisoned palladium, we get an aldehyde. Hence, B can be Ethanal, CH3CHO.
An aldehyde, on treating with dilute alkali, undergoes aldol condensation reaction. Hence, C can be CH3CH(OH)CH2CHO.
On heating an aldol product, it loses water to produce a double bond and we get CH3CH=CHCHO.
Hence, we have
संबंधित प्रश्न
Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile.
Describe the following:
Cross aldol condensation
What is substituted imine called?
Assertion: Aromatic aldehydes and formaldehyde undergo Cannizaro reaction.
Reason: Aromatic aldehydes are almost as reactive as formaldehyde.
When liquid ‘A’ is treated with a freshly prepared ammoniacal silver nitrate solution, it gives bright silver mirror. The liquid forms a white crystalline solid on treatment with sodium hydrogensulphite. Liquid ‘B’ also forms a white crystalline solid with sodium hydrogensulphite but it does not give test with ammoniacal silver nitrate. Which of the two liquids is aldehyde? Write the chemical equations of these reactions also.
Give reasons to support the answer:
Presence of Alpha hydrogen in aldehydes and ketones is essential for aldol condensation.
Cross aldol condensation occurs between
Identify A and B from the following reaction:
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\phantom{}\ce{2CH3 - C = O ->[Ba(OH)2] A ->[Δ] B + H2O}
\end{array}\]
Convert the following:
Acetaldehyde to But-2-enal
