Advertisements
Advertisements
Questions
Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
Account for the following:
The alpha (α)-hydrogens of aldehydes and ketones are acidic in nature.
Advertisements
Solution
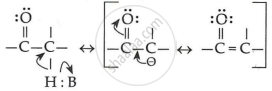
Because of the strong electron-withdrawing nature of the carbonyl groups and the resonance stabilisation of the conjugate base, aldehydes and ketones are acidic in nature.
| \[\begin{array}{cc} \ce{R}\phantom{..........}\\ \backslash\phantom{........}\\ \ce{C = O}\\ /\phantom{........}\\ \ce{H}\phantom{..........}\\ \end{array}\] |
\[\begin{array}{cc} \ce{R}\phantom{..........}\\ \backslash\phantom{........}\\ \ce{C = O}\\ /\phantom{........}\\ \ce{R}\phantom{..........}\\ \end{array}\] |
| Aldehyde | Ketone |
APPEARS IN
RELATED QUESTIONS
How will you convert ethanal into the following compound?
Butane-1, 3-diol
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile.
Cannizaro’s reaction is not given by ______.
Why is there a large difference in the boiling points of butanal and butan-1-ol?
Why are carboxylic acids more acidic than alcohols or phenols although all of them have hydrogen atom attached to a oxygen atom \[\ce{(-O-H)}\]?
Identify A and B from the following reaction:
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\phantom{}\ce{2CH3 - C = O ->[Ba(OH)2] A ->[Δ] B + H2O}
\end{array}\]
Explain Aldol condensation of ethanal.
Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound.
Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
Write a note on the aldol condensation reaction of acetaldehyde.
Which one of the following undergoes reaction with 50% sodiumhydroxide solution to give the corresponding alcohol and acid:
