Advertisements
Advertisements
प्रश्न
Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
Account for the following:
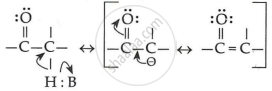
The alpha (α)-hydrogens of aldehydes and ketones are acidic in nature.
Advertisements
उत्तर
Because of the strong electron-withdrawing nature of the carbonyl groups and the resonance stabilisation of the conjugate base, aldehydes and ketones are acidic in nature.
| \[\begin{array}{cc} \ce{R}\phantom{..........}\\ \backslash\phantom{........}\\ \ce{C = O}\\ /\phantom{........}\\ \ce{H}\phantom{..........}\\ \end{array}\] |
\[\begin{array}{cc} \ce{R}\phantom{..........}\\ \backslash\phantom{........}\\ \ce{C = O}\\ /\phantom{........}\\ \ce{R}\phantom{..........}\\ \end{array}\] |
| Aldehyde | Ketone |
APPEARS IN
संबंधित प्रश्न
Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
How will you convert ethanal into the following compound?
But-2-enal
Give reasons Acetylation of aniline reduces its activation effect.
Compounds A and C in the following reaction are:
\[\ce{CH3CHO ->[(i) CH3MgBr][(ii) H2O] (A) ->[H2SO4, Δ] (B) ->[Hydroboration oxidation] (C)}\]
Assertion: Aromatic aldehydes and formaldehyde undergo Cannizaro reaction.
Reason: Aromatic aldehydes are almost as reactive as formaldehyde.
Convert the following:
Acetaldehyde to But-2-enal
Explain Aldol condensation of ethanal.
Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound.
Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
Write a note on the aldol condensation reaction of acetaldehyde.
Which one of the following undergoes reaction with 50% sodiumhydroxide solution to give the corresponding alcohol and acid:
