Advertisements
Advertisements
प्रश्न
How will you bring about the following conversion in not more than two steps?
Benzaldehyde to 3-Phenylpropan-1-ol
Advertisements
उत्तर १
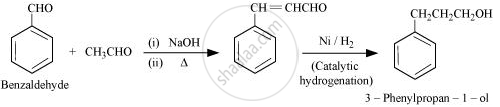
उत्तर २
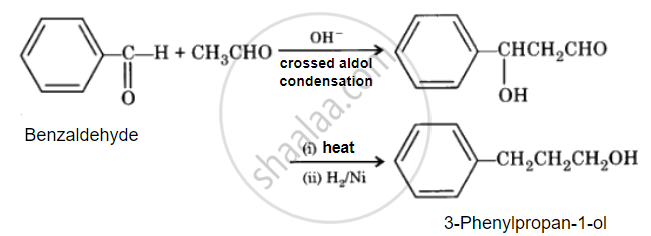
APPEARS IN
संबंधित प्रश्न
How will you convert ethanal into the following compound?
Butane-1, 3-diol
Describe the following:
Cross aldol condensation
Write a chemical equation for the following reaction:
Propanone is treated with dilute Ba( OH)2.
What is substituted imine called?
Explain aldol condensation reaction in detail.
Cannizaro’s reaction is not given by ______.
What product will be formed on reaction of propanal with 2-methylpropanal in the presence of \[\ce{NaOH}\]? What products will be formed? Write the name of the reaction also.
Assertion: Aromatic aldehydes and formaldehyde undergo Cannizaro reaction.
Reason: Aromatic aldehydes are almost as reactive as formaldehyde.
Give reasons to support the answer:
Presence of Alpha hydrogen in aldehydes and ketones is essential for aldol condensation.
\[\ce{CH3-CH2-CHO ->[dil][alkali] Product}\]
The product in the above reaction is:
Explain Aldol condensation of ethanal.
Which of the following does not give aldol condensation reaction?
Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?

Identify A and B:
When acetaldehyde is treated with dilute NaOH, the following reaction is observed.
\[\begin{array}{cc}
\ce{2CH3 - CHO ->[dil.NaOH] CH3 - CH - CH2 - CHO}\\
\phantom{...............}|\\
\phantom{.................}\ce{OH}
\end{array}\]
- What are the functional groups in the product?
- Can another product be formed during the same reaction? (Deduce the answer by doing atomic audit of reactant and product).
- Is this an addition reaction or condensation reaction?
What is aldol condensation? Explain it with suitable examples.
