Advertisements
Advertisements
प्रश्न
Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
Account for the following:
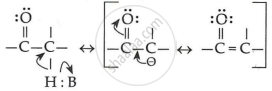
The alpha (α)-hydrogens of aldehydes and ketones are acidic in nature.
Advertisements
उत्तर
Because of the strong electron-withdrawing nature of the carbonyl groups and the resonance stabilisation of the conjugate base, aldehydes and ketones are acidic in nature.
| \[\begin{array}{cc} \ce{R}\phantom{..........}\\ \backslash\phantom{........}\\ \ce{C = O}\\ /\phantom{........}\\ \ce{H}\phantom{..........}\\ \end{array}\] |
\[\begin{array}{cc} \ce{R}\phantom{..........}\\ \backslash\phantom{........}\\ \ce{C = O}\\ /\phantom{........}\\ \ce{R}\phantom{..........}\\ \end{array}\] |
| Aldehyde | Ketone |
APPEARS IN
संबंधित प्रश्न
A compound 'A' of molecular formula C2H3OCl undergoes a series of reactions as shown below. Write the structures of A, B, C and D in the following reactions :

Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
What is meant by the following term? Give an example of the reaction in the following case.
Aldol
Which of the following compounds would undergo aldol condensation, which the Cannizzaro reaction and which neither? Write the structures of the expected products of aldol condensation and Cannizzaro reaction.
- Methanal
- 2-Methylpentanal
- Benzaldehyde
- Benzophenone
- Cyclohexanone
- 1-Phenylpropanone
- Phenylacetaldehyde
- Butan-1-ol
- 2, 2-Dimethylbutanal
Write a chemical equation for the following reaction:
Propanone is treated with dilute Ba( OH)2.
Which of the following conversions can be carried out by Clemmensen Reduction?
(i) Benzaldehyde into benzyl alcohol
(ii) Cyclohexanone into cyclohexane
(iii) Benzoyl chloride into benzaldehyde
(iv) Benzophenone into diphenyl methane
Why are carboxylic acids more acidic than alcohols or phenols although all of them have hydrogen atom attached to a oxygen atom \[\ce{(-O-H)}\]?
Assertion: Aromatic aldehydes and formaldehyde undergo Cannizaro reaction.
Reason: Aromatic aldehydes are almost as reactive as formaldehyde.
Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound.
Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
What is aldol condensation? Explain it with suitable examples.
