Advertisements
Advertisements
प्रश्न
An organic compound 'A' with the molecular formula C4H8O2 undergoes acid hydrolysis to form two compounds 'B' and 'C'. Oxidation of 'C' with acidified potassium permanganate also produces 'B'. Sodium salt of 'B' on heating with soda lime gives methane.
- Identify 'A', 'B' and 'C'.
- Out of 'B' and 'C', which will have higher boiling point? Give reason.
Advertisements
उत्तर
- A = C4H8O2 [CH3COOC2H5] ester Reactions involved are
\[\ce{\underset{A}{CH3-COOC2H5} + H2O ->[dil H2SO4] \underset{B}{CH3COOH} + \underset{C}{CH3CH2OH}}\]
Then
\[\ce{\underset{C}{CH3CH2OH} ->[KMnO4][{[O]}] \underset{B}{CH3COOH}}\]
So A = CH3COOC2H5, B = CH3COOH, C = CH3CH2OH - B has a higher boiling point than C. Because of their propensity to generate intermolecular H-bonds, carboxylic acids have higher boiling temperatures than alcohols.
APPEARS IN
संबंधित प्रश्न
A and B are two functional isomers of compound C3H6O.On heating with NaOH and I2, isomer B forms yellow precipitate of iodoform whereas isomer A does not form any precipitate. Write the formulae of A and B.
Predict the products of the following reactions :

How will you convert ethanal into the following compound?
But-2-enoic acid
Give a simple chemical test to distinguish between the following pair of compounds:
Pentan-2-one and Pentan-3-one
Alkenes decolourise bromine water in presence of CCl4 due to formation of ______.
Acetaldehyde cannot show?
Write chemical test to distinguish between the following compounds:
Phenol and Benzoic acid
The major products formed in the following reaction sequence A and B are:

Match List-I with List-II:
| List-I (Reaction) |
List-II (Reagents/Condition) |
||
| A. | 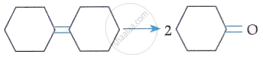 |
I. | 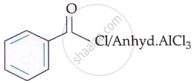 |
| B. | 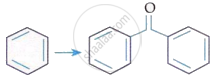 |
II. | CrO3 |
| C. | 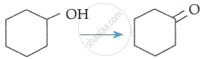 |
III. | KMnO4/KOH, Δ |
| D. | 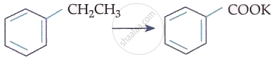 |
IV. | (i) O3 (ii) Zn-H2O |
Choose the correct answer from the options given below:
