Advertisements
Advertisements
Questions
Write the chemical equations to illustrate the following name reactions : Rosenmund reduction
Give balanced equations for the following name reactions
Rosenmund reaction
Advertisements
Solution 1
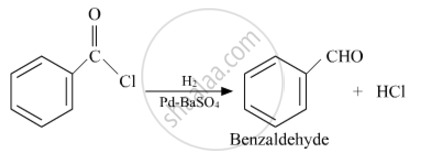
shaalaa.com
Solution 2
Rosemund reaction:
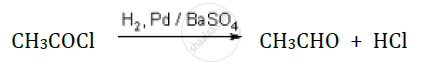
shaalaa.com
Solution 3
Rosemund reaction:
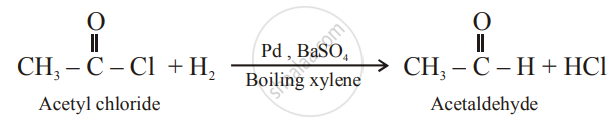
shaalaa.com
Is there an error in this question or solution?
