Definitions [18]
Primary amines (1°) are the ammonia derivatives in which one H-atom has been replaced by one alkyl or aryl group.
Tertiary amines (3°) are the ammonia derivatives in which three hydrogen atoms have been replaced by three alkyl or aryl group.
Secondary amines (2°) are the ammonia derivatives in which two hydrogen atoms have been replaced by two alkyl or aryl groups.
An amine in which three hydrogen atoms of ammonia are replaced by alkyl or aryl groups is called tertiary amine.
R–N(R′)–R″
The reaction in which amines react with acid chlorides or anhydrides to form amides is called acylation.
The method of preparation of primary amines using potassium phthalimide is called Gabriel phthalimide synthesis.
The degradation of amides to primary amines containing one carbon less is called Hofmann bromamide degradation reaction.
Organic compounds derived from ammonia by replacement of one or more hydrogen atoms by alkyl or aryl groups are called amines.
An amine in which two hydrogen atoms of ammonia are replaced by alkyl or aryl groups is called secondary amine.
R–NH–R′
The substitution reaction in which diazonium group is replaced by Cl, Br or CN in the presence of Cu(I) salts is called Sandmeyer reaction.
\[ArN_2^+Cl^-\xrightarrow{CuCl/HCl}ArCl+N_2\]
\[ArN_2^+Cl^-\xrightarrow{CuBr/HBr}ArBr+N_2\]
\[ArN_2^+Cl^-\xrightarrow{CuCN}ArCN+N_2\]
A positively charged nitrogen species formed by addition of four alkyl/aryl groups is called quaternary ammonium salt.
R₄N⁺X⁻
The reaction in which alkyl halides react with ammonia to form amines is called ammonolysis.
The reaction of amines with nitrous acid to form diazonium salts (in case of primary aromatic amines) is called diazotization reaction.
An amine in which one hydrogen atom of ammonia is replaced by an alkyl or aryl group is called primary amine.
R–NH₂
The reaction in which primary amines on heating with chloroform and alcoholic KOH form isocyanides is called Carbylamine reaction.
R – NH2 + CHCl3 + 3KOH → R – NC + 3KCl + 3H2O
The reaction of primary aromatic amines with nitrous acid at 273–278 K to form diazonium salts is called Diazotization reaction.
The reaction of primary and secondary amines with benzenesulphonyl chloride to form sulphonamides is called Hinsberg reaction.
Primary amine:
R – NH2 + C6H5SO2Cl → Sulphonamide
Secondary amine:
R2NH + C6H5SO2Cl → N,N-dialkyl sulphonamide
Tertiary amine → No reaction
The reaction in which diazonium salts couple with phenols or aromatic amines to form azo compounds is called Coupling reaction.
ArN2+ Cl− + Phenol → Azo compound
Chemica Equations [7]
\[R–NO_2\xrightarrow{H_2/Pd}R–NH_2\]
\[R\neg CN\xrightarrow{LiAlH_4}R\neg CH_2NH_2\]
R – CONH2 + Br2 + 4NaOH → R – NH2 + Na2CO3 + 2NaBr + 2H2O
R – X + NH3 → R – NH2 + HX
Salt + R – X → R – NH2
R – NH2 + R′COCl → R – NHCO – R′ + HCl
Ar – NH2 + NaNO2 + HCl → Ar – N2+ Cl−
Key Points
- Amines are derivatives of ammonia (NH₃) where one, two, or all three hydrogen atoms are replaced by alkyl or aryl groups.
- They contain a nitrogen atom with a lone pair of electrons → this lone pair makes amines basic in nature.
- Compounds of nitrogen connected to a carbonyl group are called amides (R–CO–NR'R'') — different from amines.
In amines, N atom is sp³ hybridised. Among four sp³ hybridised orbital one contains lone pair of electrons and other three contain bond pairs of electrons.
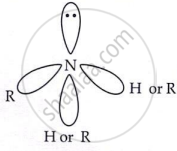
Due to a lone pair of electrons, it is pyramidal in shape.
- Amines overview: Amines are derivatives of ammonia (NH₃) where one or more H-atoms are replaced by alkyl/aryl groups; they can be saturated or unsaturated.
- Classification by source: Aliphatic amines (e.g., CH₃–NH₂, methylamine) and Aromatic amines (e.g., C₆H₅–NH₂, aniline).
- Classification by H-substitution: Primary (1°, –NH₂, e.g., methylamine), Secondary (2°, –NH–, e.g., dimethylamine), and Tertiary (3°, –N–, e.g., trimethylamine).
- Sub-types of 2° and 3° amines: Simple/symmetrical (same groups, e.g., (C₂H₅)₂NH, (C₂H₅)₃N) and Mixed/unsymmetrical (different groups, e.g., C₂H₅–NH–CH₃, C₂H₅–N(CH₃)₂).
- Examples of each class: 1° – n-butylamine, sec-butylamine, tert-butylamine, aniline; 2° – dimethylamine, diethylamine, diphenylamine; 3° – trimethylamine, triethylamine, triphenylamine.
Replace 'e' of alkane by amine'.
For example, \[ \underset{\text{Ethanamine}}{\mathrm{CH}_3\mathrm{CH}_2\mathrm{NH}_2} \quad , \quad \underset{\text{Propan-1-amine}}{\mathrm{CH}_3\mathrm{CH}_2\mathrm{CH}_2\mathrm{NH}_2} \]
Secondary amines are named as N-alkylaminoalkanes, and tertiary amines are named as N, N-dialkylaminoalkanes
For example,
\[ \underset{\text{N-methylethanamine}}{\mathrm{CH}_3 - \underset{\underset{\displaystyle \mathrm{H}}{|}}{\mathrm{N}} - \mathrm{CH}_2 - \mathrm{CH}_3} \quad , \quad \underset{\text{N-methylmethanamine}}{\mathrm{CH}_3 - \underset{\underset{\displaystyle \mathrm{H}}{|}}{\mathrm{N}} - \mathrm{CH}_3} \]
Aromatic amine,
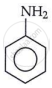
It is named as aniline or benzenamine.
- Common system: Aliphatic amine is named by adding 'amine' to the alkyl group (e.g., CH₃NH₂ – methylamine).
- Same groups in 2°/3° amines: Use prefix di or tri before the alkyl group name (e.g., trimethylamine).
- IUPAC – Primary amines: Named as alkanamines, replacing 'e' of alkane with 'amine' (e.g., methanamine).
- IUPAC – 2° & 3° amines: Named as N-alkylalkanamine or N,N-dialkylalkanamine (e.g., N,N-dimethylmethanamine).
- Multiple —NH₂ groups: Positions are numbered, terminal 'e' retained (e.g., ethane-1,2-diamine); aromatic amines named as aniline derivatives (e.g., N, N-dimethylaniline).
Reduction of Alkyl Nitriles (Nitrile Reduction):
\[ \mathrm{R} - \mathrm{C} \equiv \mathrm{N} \xrightarrow[\mathrm{LiAlH}_4 \text{ or } \mathrm{Na} / \mathrm{C}_2\mathrm{H}_5\mathrm{OH}]{\text{Raney Ni} / \mathrm{H}_2} \mathrm{R} - \mathrm{CH}_2\mathrm{NH}_2 \]
Reduction of Amides:
\[ \mathrm{R} - \mathrm{CONH}_2 \xrightarrow{\mathrm{LiAlH}_4 / \text{ether}} \mathrm{R} - \mathrm{CH}_2\mathrm{NH}_2 \]
Reduction of Oximes:
\[ \mathrm{R} - \mathrm{CH} = \mathrm{NOH} + 4[\mathrm{H}] \xrightarrow[\mathrm{LiAlH}_4]{\mathrm{Na} / \mathrm{C}_2\mathrm{H}_5\mathrm{OH}} \mathrm{R} - \mathrm{CH}_2\mathrm{NH}_2 + \mathrm{H}_2\mathrm{O} \]
Reductive Amination (from Aldehydes/Ketones):
\[ \mathrm{R} - \mathrm{CH} = \mathrm{O} \xrightarrow{\mathrm{NH}_2\mathrm{OH}} \mathrm{R} - \mathrm{CH} = \mathrm{N} - \mathrm{OH} \xrightarrow{\mathrm{LiAlH}_4} \mathrm{R} - \mathrm{CH}_2 - \mathrm{NH}_2 \]
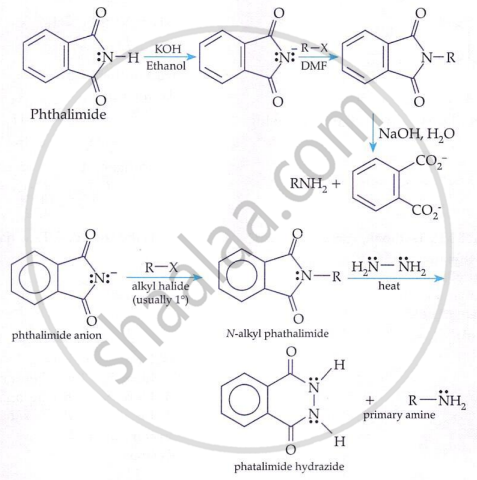
Gabriel Phthalimide Reaction (for 1° amines only):
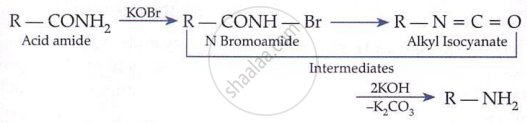
Hoffmann Bromamide Degradation (gives 1° amine with one less carbon):
\[ \underset{\text{Amide}}{\mathrm{RCONH}_2} + \mathrm{Br}_2 + 4\mathrm{KOH} \longrightarrow \underset{1^\circ \text{ amine}}{\mathrm{RNH}_2} + 2\mathrm{KBr} + 2\mathrm{H}_2\mathrm{O} + \mathrm{K}_2\mathrm{CO}_3 \]
Mechanism:
- Boiling point vs non-polar compounds: Due to intermolecular H-bonding, amines boil at a higher temperature than non-polar compounds such as hydrocarbons.
- Boiling point vs alcohols/acids: The boiling points of amines are less than those of alcohols and carboxylic acids.
- Solubility of lower amines: Lower members of aliphatic amines are soluble in water due to intermolecular H-bonding with water.
- Effect of alkyl group size: Solubility in water decreases as the size of the alkyl group increases.
- Order of boiling points & aromatic amines: Propane < ethylamine < diethylamine < n-butylamine < ethyl alcohol < propanoic acid; aromatic amines are insoluble in water due to the bulky size of the phenyl group.
- Amines are basic due to the lone pair on nitrogen → can donate to a proton (Bronsted base) or Lewis acids.
- In gas/non-aqueous phase: Basicity ∝ +I effect → 3° > 2° > 1° > NH₃
- In aqueous phase: Solvation (hydration) also plays a role.
Aqueous basicity order (aliphatic):
\[ \underset{(2^\circ)}{(\mathrm{CH}_3)_2\mathrm{NH}} > \underset{(1^\circ)}{\mathrm{CH}_3\mathrm{NH}_2} > \underset{(3^\circ)}{(\mathrm{CH}_3)_3\mathrm{N}} > \mathrm{NH}_3 \]
Only 1° amines (aliphatic and aromatic) react with CHCl₃ and alc. KOH to give isocyanides (carbylamines) with very bad smell.
- Used as a test for 1° amines (Carbylamine test / Isocyanide test).
- 2° and 3° amines do NOT give this reaction.
\[ \underset{1^\circ \text{ amine}}{\mathrm{RNH}_2} + \mathrm{CHCl}_3 + \text{Alc. } 3\mathrm{KOH} \longrightarrow \underset{\text{Alkyl isocyanide}}{\mathrm{RNC}} + 3\mathrm{KCl} + 3\mathrm{H}_2\mathrm{O} \]
HNO₂ is generated in situ: NaNO₂ + HCl → HNO₂
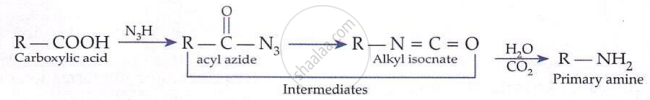
Primary Aliphatic Amines:
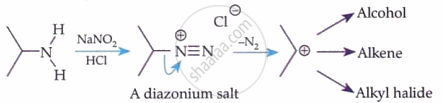
Primary Aromatic Amines (Diazotisation):
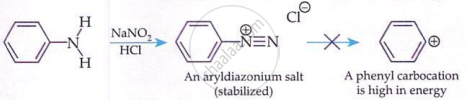
'Aromatic diazonium salts are stable due to delocalisation of the +ve charge in the benzene ring.
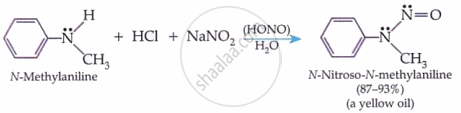
Secondary Amines (both aliphatic and aromatic):
\[ \underset{\text{Dimethylamine}}{(\mathrm{CH}_3)_2 \ddot{\mathrm{N}}\mathrm{H}} + \mathrm{HCl} + \mathrm{NaNO}_2 \xrightarrow{\mathrm{H}_2\mathrm{O}} [(\mathrm{HONO})] \underset{\substack{\text{N-Nitrosodimethylamine} \\ \text{(a yellow oil)}}}{(\mathrm{CH}_3)_2 \ddot{\mathrm{N}} - \ddot{\mathrm{N}} = \mathrm{O}} \]
Tertiary Aliphatic Amines:
\[ \underset{\text{Tertiary aliphatic amine}}{2\mathrm{R}_3\mathrm{N}} + \mathrm{HX} + \mathrm{NaNO}_2 \rightleftharpoons \underset{\text{Amine salt}}{\mathrm{R}_3\overset{+}{\mathrm{N}}\mathrm{H} \text{ } \mathrm{X}^-} + \underset{\text{N-Nitrosoammonium compound}}{\mathrm{R}_3\overset{+}{\mathrm{N}}-\ddot{\mathrm{N}}=\mathrm{O}\mathrm{X}^-} \]
Tertiary Aromatic Amines:
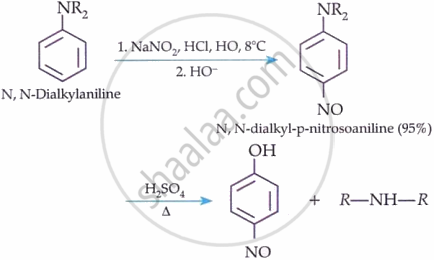
Electrophilic substitution reaction of arylamines:
- Diazonium salts: Compounds with the general formula ArN₂⁺X⁻ (e.g., C₆H₅N₂⁺Cl⁻ = Benzenediazonium chloride).
- X⁻ = Cl⁻, Br⁻, HSO₄⁻, NO₃⁻, BF₄⁻, etc.
- Benzenediazonium chloride: Colourless crystalline solid; readily soluble in water; stable in cold but decomposes on warming; decomposes easily in dry state.
- Benzenediazonium fluoroborate: Water insoluble, stable at room temperature.
- The stability of arenediazonium ions is due to resonance — the positive charge is delocalised into the benzene ring.
Primary amines are prepared by treating potassium phthalimide with alkyl halides followed by hydrolysis.
Key points:
-
Gives only primary amines.
-
Not suitable for aryl halides.
-
Avoids formation of secondary and tertiary amines.
Primary and secondary amines react with acid chlorides or anhydrides to form amides via nucleophilic substitution.
Key Points:
-
Used for identification of amines.
-
Tertiary amines do not undergo acylation.
-
Reaction carried out in presence of base.
Primary amides on treatment with bromine and sodium hydroxide give primary amines containing one carbon atom less than the parent amide.
Key Points:
-
Carbonyl carbon is lost as CO₂.
-
Rearrangement reaction.
-
Useful for chain shortening.
Amines are basic in nature due to the presence of a lone pair of electrons on nitrogen which can accept a proton.
Factors affecting basicity:
- +I effect of alkyl groups increases basicity.
- Solvation effect in aqueous solution.
- Steric hindrance decreases basicity.
- Aromatic amines are less basic due to resonance.
Order in gaseous phase:
3∘ > 2∘ >1∘ >NH3
Order in aqueous phase:
2∘ > 1∘ > 3∘ > NH3
Important Questions [102]
- Give the Structures of A, B and C in the Following Reactions :
- Give the Structures of A, B and C in the Following Reactions :
- Write Structures of Compounds a and B in Each of the Following Reactions:
- Among the following, which is the strongest base? [1]
- Arrange the following: In increasing order of solubility in water: C6H5NH2, (C2H5)2NH, C2H5NH2
- Give Reasons for the Following: Ch3nh2 is More Basis than C6h5nh2.
- Write the Iupac Name of the Given Compound :
- Give Reasons Although –Nh2 is O/P Directing Group, Yet Aniline on Nitration Gives a Significant Amount of M-nitroaniline
- Do the Following Conversions in Not More than Two Steps : Propanone to Propene
- Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine. CH3NHCH(CH3)2
- Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine. (CH3CH2)2NCH3
- Account for the following: pKb of aniline is more than that of methylamine.
- Account for the following: Methylamine in water reacts with ferric chloride to precipitate hydrated ferric oxide.
- Do the following conversions in not more than two steps:
- Do the following conversions in not more than two steps: CHA3CN to CHA3−C−CHA3...........||...........O
- How will you convert ethanoic acid into methanamine?
- Do the Following Conversions in Not More than Two Steps : Ethyl Benzene to Benzoic Acid
- Give simple chemical tests to distinguish between the following pair of compounds: Benzaldehyde and Benzoic acid
- An organic compound (A) with molecular formula C3H7NO on heating with Br2 and KOH forms a compound (B). Compound (B) on heating with CHCl3 and alcoholic KOH produces a foul-smelling compound (C)
- Using IUPAC norms, write the formula of Hexaamminecobalt(III) sulphate.
- Write the Structure of 2,4-dinitrochlorobenzene
- Give the Structures of A, B and C in the Following Reactions :
- Arrange the Following in the Increasing Order of Their Pkb Values: C6h5nh2, C2h5nh2, C6h5nhch3
- Give the Structures of A, B and C in the Following Reactions :
- Illustrate the Following Reaction Giving Suitable Example in Each Case: Gabriel Phthalimide Synthesis
- Give reasons for the following: Ammonolysis of alkyl halides is not a good method to prepare pure primary amines.
- Amides can be converted into amines by the reaction named ______.
- A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of
- An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions
- Write the chemical equation involved in the following reaction: Hoffmann-bromamide degradation reaction
- Write Structures of Compounds a and B in Each of the Following Reactions:
- How do you convert the following: C6H5CONH2 to C6H5NH2
- How do you convert the following: Ethanenitrile to ethanamine
- Gabriel Phthalimide Synthesis Not Prefered for Preparing Aromatic Primary Amines Because Amine Formation Involves Nucleophilic Substituion 2 ( ) Sn of Alkyl Halides by the Anion Formed by
- Which of the following would not be a good choice for reducing nitrobenzene to aniline?
- Give the Structures of A, B and C in the Following Reactions
- Give the structures of A, B and C in the following reactions
- Give the Structures of A, B and C in the Following Reactions :
- Arrange the Following in Increasing Order of Basic Strength : C6h5nh2, C6h5nhch3, C6h5n(Ch3)2
- Give Reasons Ch3nh2 is More Basic than C6h5nh2.
- Arrange the following in increasing order of their boiling point: C2H5OH, C2H5NH2, (C2H5)3N
- Arrange the Following in Increasing Order of Their Basic Strength
- Arrange in increasing order of basic strength C6H5 – NH2, C6H5 – CH2 – NH2, C6H5 – NH – CH3
- Give reasons for the following: (CH3)2NH is more basic than (CH3)3N in an aqueous solution.
- Arrange the following: C2H5NH2, C2H5OH, (CH3)3N – in the increasing order of their boiling point
- Illustrate the Following Reactions Giving Suitable Example in Each Case Ammonolysis
- Give Reason (Ch3)2nh is More Basic than (Ch3)3n in an Aqueous Solution
- Arrange the Following in the Increasing Order of Their Pkb Values:
- Give reason for the following: Primary amines have higher boiling point than tertiary amines.
- Arrange the following in increasing order of their basic strength in aqueous solution: CHA3NHA2,(CHA3)A3N,(CHA3)A2NH
- Arrange the Following: Aniline, P-nitroaniline, P-methylaniline - in the Increasing Order of Their Basic Strength
- Write the Structures of Main Products When Aniline Reacts with the Following Reagents : HCl
- Write the chemical equations involved when aniline is treated with the following reagents: HCI
- Why does NH3 act as a Lewis base?
- Arrange the following: In increasing order of basic strength: C6H5NH2, C6H5NHCH3, C6H5CH2NH2
- Write the Structures of the Main Products of the Following Reactions: -1
- Write the Structures of the Main Products of the Following Reactions: - 2
- Write the Structures of the Main Products of the Following Reactions: 3
- Account for the following: Arrange the following compounds in the increasing order of their basic strength in aqueous solution: CH3NH2,(CH3)3N,(CH3)2NH
- Among the following, which has the highest value of pKb?
- Write the Equation Involved in Friedal-crafts Alkylation of Phenol
- Out of Sn1 and Sn2, Which Reaction Occurs with (A) Inversion of Configuration (B) Racemisation
- What is the role of pyridine in the acylation reaction of amines?
- Which Alkyl Halide from the Following Pair is Chiral and Undergoes Faster Sn2 Reaction?
- Orange Colour of `Cr_2o_7^(2–)` Ion Changes to Yellow When Treated with an Alkali. Why?
- Write Equations of the Following Reactions: Carbyl Amine Reaction
- Give a Simple Chemical Test to Distinguish Between Aniline and N,N-dimethylaniline
- Write the Chemical Equations Involved in the Following Reactions: Carbylamine Reaction
- Write the chemical equations involved when aniline is treated with the following reagents: CHCI3 + KOH
- Write the structures of A, B and C in the following reaction:
- Draw the Structures of the Following Compounds : N2o5
- What is Hinsberg's reagent?
- On Reaction with Benzene Sulphonyl Chloride, Primary Amine Yields Product Soluble in Alkali Whereas Secondary Amine Yields Product Insoluble in Alkali.
- Give a Simple Chemical Test to Distinguish Between the Following Pair of Compounds : (CH3)2NH and (CH3)3N
- Write Chemical Equation in Support of Your Answer. Out of Cl and O2n Cl. Which One is More Reactive
- A primary amine 'A 'C2H7N reacts with alkyl halide (C2H5l) to give secondary amine 'B'. 'B' reacts with C6H5SO2Cl to give a solid 'C' which is insoluble in alkali. Identify 'A', 'B', 'C'
- Give reasons for the following: Aniline does not undergo Friedel- Crafts reaction.
- Write the Structures of Main Products When Aniline Reacts with the Following Reagents : (CH3CO)2O/pyridine
- Assertion (A): Bromination of benzoic acid, gives m-bromobenzoic acid. Reason (R): Carboxyl group increases the electron density at the meta position.
- Write the chemical equations involved when aniline is treated with the following reagents: Br2 water
- Write the structures of main products when aniline reacts with the following reagents : Br2 water
- Illustrate the Following Reactions Giving Suitable Example in Each Case Acetylation of Amines
- How can the activating effect of the −NH2 group in aniline be controlled?
- Account for the following: Although the amino group is o, p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
- Aniline does not give Friedel-Crafts reaction. Give a reason.
- How Will You Convert the Following? Aniline into N−Phenylethanamide
- Illustrate the following reaction giving suitable example in each case : Diazotisation
- Write Equations of the Following Reactions: Acetylation of Aniline
- Give Reason Aromatic Diazonium Salts Are More Stable than Aliphatic Diazonium Salts.
- Write the Structures of Main Products When Benzene Diazonium Chloride Reacts with the Following Reagents H2O
- Write the Structures of Main Products When Benzene Diazonium Chloride
- Write the structures of A, B and C in the following reaction: N2+Cl- ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C
- Write the Structures of Main Products When Benzene Diazonium Chloride Reacts with the Following Reagents: CuCN/KCN
- Write the Structures of A, B and C in the Following
- Illustrate Sandmeyer's reaction with an equation.
- Give the Structures of A, B and C in the Following Reaction
- Give the Structures of A, B and C in the Following Reactions
- Illustrate the Following Reactions Giving Suitable Example in Each Case Coupling Reaction
- When benzene diazonium chloride reacts with phenol, it forms a dye. This reaction is called ______.
- Write Equations of the Following Reactions: Coupling Reaction
- Write the Major Products(S) in the Following
- An Aromatic Compound 'A' on Heating with Br2 and Koh Forms a Compound 'B' of Molecular Formula C6h7n Which on Reacting with Chcl3 and Alcoholic Koh Produces a Foul-smelling Compound 'C'
Concepts [19]
- Concept of Amines
- Structure of Amines
- Classification of Amines
- Nomenclature of Amines
- Preparation of Amines
- Physical Properties of Amines
- Chemical Reactions of Amines - Basic Character of Amines
- Chemical Reactions of Amines - Alkylation and Acylation
- Chemical Reactions of Amines - Carbylamine Reaction
- Chemical Reactions of Amines - Reaction with Nitrous Acid
- Chemical Reactions of Amines - Reaction with Arylsulphonyl Chloride
- Chemical Reactions of Amines - Electrophilic Substitution
- Diazonium Salts
- Method of Preparation of Diazonium Salts
- Physical Properties of Diazonium Salts
- Chemical Reaction of Diazonium Salts - Reactions Involving Displacement of Nitrogen
- Chemical Reaction of Diazonium Salts - Reactions Involving Retention of Diazo Group
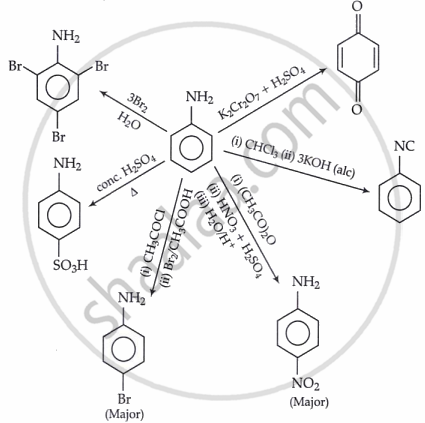
- Importance of Diazonium Salts in Synthesis of Aromatic Compounds
- Overview of Amines
