Advertisements
Advertisements
Question
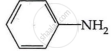
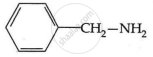
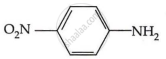
Among the following, which has the highest value of pKb?
Options
Advertisements
Solution
Explanation:
The pKb number is proportional to the basic strength. The electron density on the nitrogen atom drops due to the presence of the electron-withdrawing NO2 group.
As a result, phenylmethanamine is the least basic. Thus, it will have the highest pKb value.
APPEARS IN
RELATED QUESTIONS
Arrange the following:
Aniline, p-nitroaniline, p-methylaniline - in the increasing order of their basic strength
Arrange the following:
In decreasing order of the pKb values:
C2H5NH2, C6H5NHCH3, (C2H5)2NH and C6H5NH2
Arrange the following:
In increasing order of basic strength:
C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2
Arrange the following:
In decreasing order of basic strength in the gas phase:
C2H5NH2, (C2H5)2NH, (C2H5)3N and NH3
Write the structures of the main products of the following reactions:

Account for the following:
Acylation of aniline is carried out in the presence of pyridine.
What is the correct decreasing order of the basic character of the three amines and ammonia?
Give reasons for the following observation:
pKb of aniline is lower than the m-nitroaniline.
Account for the following:
Arrange the following compounds in the increasing order of their basic strength in aqueous solution: CH3NH2,(CH3)3N,(CH3)2NH
State one reason for the following:
Alkylamine is soluble in water, whereas arylamine is insoluble in water.