Advertisements
Advertisements
Question
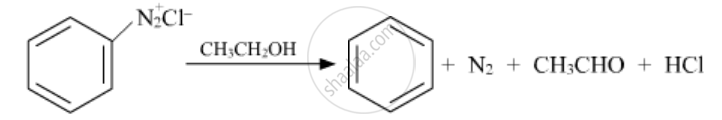
Write the structures of the main products of the following reactions:

Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
Write the structures of main products when aniline reacts with the following reagents : HCl
Arrange the following:
In decreasing order of the pKb values:
C2H5NH2, C6H5NHCH3, (C2H5)2NH and C6H5NH2
Arrange the following:
In increasing order of basic strength:
C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2
Write the structures of the main products of the following reactions:

Arrange the following in decreasing order of their basic strength:
C6H5NH2, C2H5NH2, (C2H5)2NH2, NH3
The correct increasing order of basic strength for the following compounds is ______.
(I)

(II)

(III)

When methyl iodide is heated with ammonia, what is the product obtained?
When ethanol is mixed with ammonia and passed over alumina the compound formed is which compound?
State one reason for the following:
Alkylamine is soluble in water, whereas arylamine is insoluble in water.
The correct order of the increasing basic nature of Ammonia, Methylamine and Aniline is:
