Advertisements
Advertisements
Questions
Account for the following:
Although the amino group is o, p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
Give reasons for the following:
Although the –NH2 group is o/p directing in an electrophilic substitution reaction, yet aniline on nitration gives a good yield of m-nitroaniline.
Advertisements
Solution 1
Nitration is carried out in an acidic medium. In an acidic medium, aniline is protonated to give anilinium ions (which are meta-directing).
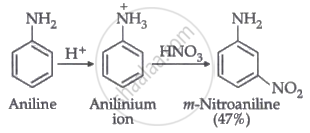
For this reason, aniline on nitration gives a substantial amount of m-nitroaniline.
Solution 2
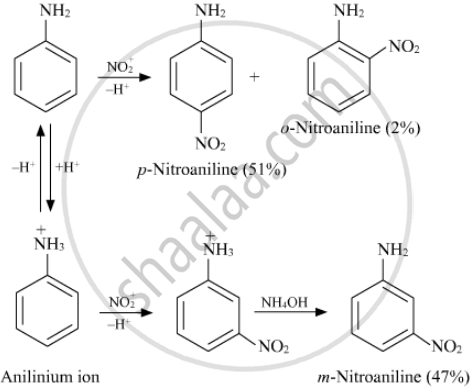
Nitration is usually carried out with a mixture of conc. HNO3 + conc. H2SO4. In the presence of these acids, most of the aniline gets protonated to form an anilinium ion. Therefore, in the presence of acids, the reaction mixture consists of aniline and anilinium ions. Now, the –NH2 group in aniline is activating and p-directing, while the \[\ce{-NH+3}\] group in the anilinium ion is deactivating and m-directing. Nitration of aniline (due to steric hindrance at the o-position) mainly gives p-nitroaniline; the nitration of anilinium ions gives m-nitroaniline. In actual practice, approx. a 1 : 1 mixture of p-nitroaniline and m-nitroaniline is obtained. Thus, nitration of aniline gives a substantial amount of m-nitroaniline due to protonation of the amino group.
APPEARS IN
RELATED QUESTIONS
How is chlorobenzene prepared from aniline?
What is the action of acetic anhydride on ethylamine?
What is the action of the following reagents on aniline?
Bromine water
Write the structures of main products when aniline reacts with the following reagents :
(CH3CO)2O/pyridine
Write short notes on acetylation.
What is the action of acetic anhydride on diethylamine?
What is the action of the following reagents on aniline?
Acetic anhydride
How will you convert the following?
Aniline into N−phenylethanamide
In the nitration of benzene using a mixture of conc. \[\ce{H2SO4}\] and conc. \[\ce{HNO3}\], the species which initiates the reaction is ______.
What is the role of \[\ce{HNO3}\] in the nitrating mixture used for nitration of benzene?
Assertion: N, N-Diethylbenzene sulphonamide is insoluble in alkali.
Reason: Sulphonyl group attached to nitrogen atom is strong electron-withdrawing group.
When bromination of aniline is carried out by protecting – NH2. The major product is
Give reasons for the following observation:
Aniline is acetylated before nitration reaction.
In the following reaction, the reason for why meta-nitro product also formed is:

Aniline does not give Friedel-Crafts reaction. Give a reason.
Given below are two statements:
Statement I: Aniline does not undergo a Friedel-Crafts alkylation reaction.
Statement II: Aniline cannot be prepared through Gabriel synthesis.
In the light of the above statements, choose the correct answer from the options given below:
Identify the major product C formed in the following reaction sequence:
\[\ce{CH3 - CH2 - CH2 - I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH][Br2] \underset{(major)}{C}}\]
