Advertisements
Advertisements
Question
Aniline does not give Friedel-Crafts reaction. Give a reason.
Advertisements
Solution
Aniline does not undergo Friedel-Craft's reaction because the reaction takes place in the presence of AlCl3, but AlCl3 is a Lewis acid, whereas aniline is a strong basic. Thus, aniline interacts with AlCl3 to produce a salt.
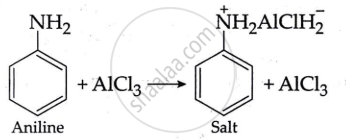
The positive charge on the N-atom prevents electrophilic substitution in the benzene ring. As a result, aniline does not undergo the Friedel-Craft reaction.
APPEARS IN
RELATED QUESTIONS
Give reasons for the following:
Aniline does not undergo Friedel- Crafts reaction.
Account for the following:
Although the amino group is o, p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
Write short notes on acetylation.
What is the action of acetic anhydride on diethylamine?
In the nitration of benzene using a mixture of conc. \[\ce{H2SO4}\] and conc. \[\ce{HNO3}\], the species which initiates the reaction is ______.
What is the role of \[\ce{HNO3}\] in the nitrating mixture used for nitration of benzene?
A solution contains 1 g mol. each of p-toluene diazonium chloride and p-nitrophenyl diazonium chloride. To this 1 g mol. of alkaline solution of phenol is added. Predict the major product. Explain your answer.
How can the activating effect of the −NH2 group in aniline be controlled?
Given below are two statements:
Statement I: Aniline does not undergo a Friedel-Crafts alkylation reaction.
Statement II: Aniline cannot be prepared through Gabriel synthesis.
In the light of the above statements, choose the correct answer from the options given below:
Identify the major product C formed in the following reaction sequence:
\[\ce{CH3 - CH2 - CH2 - I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH][Br2] \underset{(major)}{C}}\]
