Advertisements
Advertisements
Question
A solution contains 1 g mol. each of p-toluene diazonium chloride and p-nitrophenyl diazonium chloride. To this 1 g mol. of alkaline solution of phenol is added. Predict the major product. Explain your answer.
Advertisements
Solution
This reaction is an example of electrophilic aromatic substitution. In alkaline medium, phenol forms phenoxide ion which is more electron-rich than phenol and hence more reactive for electrophilic attack. The electrophile in this reaction is aryl diazonium cation. p-Nitrophenyldiazonium cation is a stronger electrophile than p-toluene diazonium cation. Therefore, it couples preferentially with phenol.
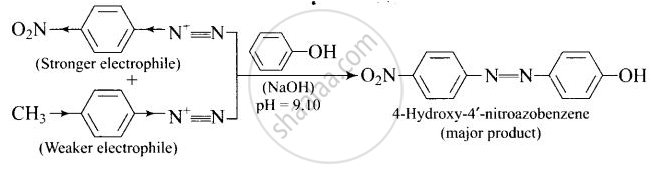
APPEARS IN
RELATED QUESTIONS
How is chlorobenzene prepared from aniline?
What is the action of acetic anhydride on ethylamine?
What is the action of the following reagents on aniline?
Bromine water
Give reasons for the following:
Aniline does not undergo Friedel- Crafts reaction.
Write the chemical equations involved when aniline is treated with the following reagents:
Br2 water
What is the action of acetic anhydride on diethylamine?
What is the action of the following reagents on aniline?
Acetic anhydride
What is the action of the following reagents on aniline?
Hot and conc. sulphuric acid
Illustrate the following reactions giving suitable example in each case
Acetylation of amines
How will you convert the following?
Aniline into N−phenylethanamide
In the nitration of benzene using a mixture of conc. \[\ce{H2SO4}\] and conc. \[\ce{HNO3}\], the species which initiates the reaction is ______.
Assertion: N, N-Diethylbenzene sulphonamide is insoluble in alkali.
Reason: Sulphonyl group attached to nitrogen atom is strong electron-withdrawing group.
When bromination of aniline is carried out by protecting – NH2. The major product is
How can the activating effect of the −NH2 group in aniline be controlled?
Assertion (A): Bromination of benzoic acid, gives m-bromobenzoic acid.
Reason (R): Carboxyl group increases the electron density at the meta position.
Given below are two statements:
Statement I: Aniline does not undergo a Friedel-Crafts alkylation reaction.
Statement II: Aniline cannot be prepared through Gabriel synthesis.
In the light of the above statements, choose the correct answer from the options given below:
Identify the major product C formed in the following reaction sequence:
\[\ce{CH3 - CH2 - CH2 - I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH][Br2] \underset{(major)}{C}}\]
