Advertisements
Advertisements
Question
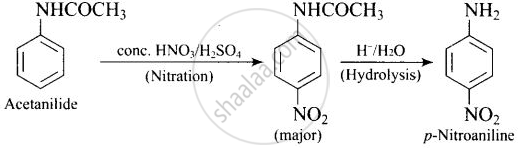
Write following conversions:
acetanilide `->` p-nitroaniline
Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
How are propan-1-amine and propan-2-amine prepared from oxime?
Write a short note on Hoffmann bromamide degradation.
Identify the compounds 'A' and 'B' in the following equation:

Write a short note on the following:
Hoffmann’s bromamide reaction
Write the reaction of aromatic primary amine with nitrous acid.
Name the process of breaking C-X bond by ammonia in preparation of amines.
Identify the product obtained, when benzamide is treated with bromine and aqueous sodium hydroxide.
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
In aqueous phase the order of basic strength of alkylamine is ______.
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Amongst the following, the strongest base in aqueous medium is ______.
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
How will you carry out the following conversion?

How will you carry out the following conversions?

A primary amine is formed by an amide on treatment with bromine and alkali. The primary amine has
Ethylamine can be prepared by the action of bromine and caustic potash on which compound?
Write a short note on Ammonolysis.
Write short notes on the following:
Ammonolysis
