Advertisements
Advertisements
Question
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
Options
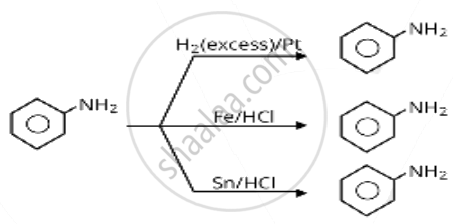
\[\ce{H2 (excess)/Pt}\]
\[\ce{LiAlH4}\] in ether
\[\ce{Fe}\] and \[\ce{HCl}\]
\[\ce{Sn}\] and \[\ce{HCl}\]
Advertisements
Solution
\[\ce{LiAlH4}\] in ether
Explanation:
Lithium aluminium hydride in either is a strong reducing agent that donated its \[\ce{H-}\] hydride ion to any \[\ce{C = O}\] containing a functional group. In addition to \[\ce{LiAlH}\], to aryl nitro compounds, no reaction will be observed, the desired products of amines will not be produced, rather it will form diazobenzene products.
\[\ce{2C4H5NO ->[LiAlH4/ether] C6H5N = N - C6H5}\]
APPEARS IN
RELATED QUESTIONS
Give the structures of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[HNO2][273 K] B ->[C6H5OH] C}\]
Write the reaction of aliphatic primary amine with nitrous acid.
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Write structures of compounds A and B in each of the following reactions:
Answer the following
Explain Gabriel phthalimide synthesis.
Write reactions to bring about the following conversions.
Acetamide to Ethylamine
Write reactions to bring about the following conversions.
Acetamide to methylamine
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Identify the product 'A' in the following reaction.
\[\ce{Aniline ->[(CH3CO)2O][Pyridine] A}\]
Alkyl cyanides on reduction by sodium and ethanol give primary amines. This reaction is called as ____________.
Which of the following reagents is used in Hofmann's elimination reaction of amines?
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds:
Identify A and B in the following reaction.

How will you carry out the following conversion?

Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Acetamide and ethyl amide can be distinguished by reacting with.
Write short note on the following:
Ammonolysis
Write a short note on Ammonolysis.
Assertion: Amimonolysis of alkyl halides involves the reaction between alkyl halides and alcoholic ammonia.
Reason: Ammonolysis of alkyl halides produces secondary amines only.
