Advertisements
Advertisements
Question
Assertion: Aromatic 1° amines can be prepared by Gabriel Phthalimide Synthesis.
Reason: Aryl halides undergo nucleophilic substitution with anion formed by phthalimide.
Options
Both assertion and reason are wrong.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Assertion is correct statement but reason is wrong statement.
Both assertion and reason are correct statements and reason is correct explanation of assertion.
Assertion is wrong statement but reason is correct statement.
Advertisements
Solution
Both assertion and reason are wrong.
Explanation:
Aromatic amines are never obtained by Gabriel phthalimide synthesis.
APPEARS IN
RELATED QUESTIONS
Write the reaction of aromatic primary amine with nitrous acid.
Identify 'A' and 'B' in the following reaction and rewrite the complete reaction :

Give the structures of A, B and C in the following reactions :
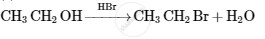
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
Identify the product 'A' in the following reaction.
\[\ce{Aniline ->[(CH3CO)2O][Pyridine] A}\]
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
Identify the product obtained when benzamide is treated with bromine and aqueous sodium hydroxide?
Which of the following amines cannot be prepared by Gabriel phthalimide synthesis?
Which of the following compounds is the weakest Brönsted base?
Among the following amines, the strongest Brönsted base is:
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
Which of the following CANNOT be prepared by ammonolysis of alkyl halide?
Give reasons for the following:
Ammonolysis of alkyl halides is not a good method to prepare pure primary amines.
Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
The amine 'A' when treated with nitrous acid gives yellow oily substance. The amine A is ______.
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Write a short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis
Write short note on the following:
Ammonolysis
