Advertisements
Advertisements
Question
Among the following amines, the strongest Brönsted base is:
Options
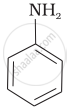
\[\ce{NH3}\]
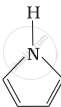
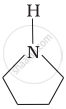
Advertisements
Solution

Explanation:
Pyrrolidine is the strongest of two bases because the lone pair of nitrogen does not involve sin resonance and the presence of two alkyl basic compounds increases the basic strength among the given four compounds.
APPEARS IN
RELATED QUESTIONS
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Give the structures of A, B and C in the following reactions :

Accomplish the following conversion:
Benzyl chloride to 2-phenylethanamine
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2Br ->[KCN] A ->[LiAlH4] B ->[HNO2][0^\circ C] C}\]
Choose the most correct option.
Which of the following compounds will dissolve in aqueous NaOH after undergoing reaction with Hinsberg reagent?
Answer in one sentence.
Which amide does produce ethanamine by Hofmann bromamide degradation reaction?
Answer the following
Explain the ammonolysis of alkyl halides.
Mendius reaction is used to convert _____________
Write reactions to bring about the following conversions.
Acetamide to methylamine
Quaternary ammonium salt is formed:
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanamine is ______.
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(i) Acetyl chloride/pyridine followed by reaction with conc.\[\ce{H2SO4 }\] + conc. \[\ce{HNO3}\].
(ii) Acetic anyhdride/pyridine followed by conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iii) Dil. HCl followed by reaction with conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iv) Reaction with conc.\[\ce{HNO3}\] + conc.\[\ce{H2 SO4}\].
How will you carry out the following conversion?

The Gabriels' phthalimide synthesis is used in the synthesis of
Which of the following reactions will not give a primary amine?
Which of the following compound gives pink colour on reaction with phthalic anhydride in cone. H2SO4 followed by treatment with NaOH?
Which of the following amines can be prepared by Gabriel phthalimide reaction?
Write a short note on the following:
Ammonolysis
