Advertisements
Chapters
2: Solutions
3: Electrochemistry
4: Chemical Kinetics
5: Surface Chemistry
6: General Principle and Processes of Isolation of Elements
7: The p-block Elements
8: The d-and f-Block Elements
9: Coordination Compounds
10: Haloalkanes and Haloarenes
11: Alcohols, Phenols and Ethers
12: Aldehydes, Ketones and Carboxylic Acids
▶ 13: Amines
14: Biomolecules
15: Polymers
16: Chemistry In Everyday Life
![NCERT Exemplar solutions for Chemistry Exemplar [English] Class 12 chapter 13 - Amines NCERT Exemplar solutions for Chemistry Exemplar [English] Class 12 chapter 13 - Amines - Shaalaa.com](/images/chemistry-exemplar-english-class-12_6:ff5a05b4361741f5816a7388844a6202.png)
Advertisements
Solutions for Chapter 13: Amines
Below listed, you can find solutions for Chapter 13 of CBSE, Karnataka Board PUC NCERT Exemplar for Chemistry Exemplar [English] Class 12.
NCERT Exemplar solutions for Chemistry Exemplar [English] Class 12 13 Amines Multiple Choice Questions (Type - I) [Pages 180 - 193]
Which of the following is a 3° amine?
1-methylcyclohexylamine
Triethylamine
tert-butylamine
N-methylaniline
The correct IUPAC name for \[\ce{CH2 = CHCH2 NHCH3}\] is ______.
Allylmethylamine
2-amino-4-pentene
4-aminopent-1-ene
N-methylprop-2-en-1-amine
Amongst the following, the strongest base in aqueous medium is ______.
\[\ce{CH3NH2}\]
\[\ce{NCCH2NH2}\]
\[\ce{(CH3)2NH}\]
\[\ce{C6H5NHCH3}\]
Which of the following is the weakest Brönsted base?
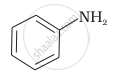
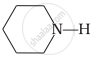
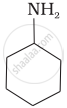
\[\ce{CH3NH2}\]
Benzylamine may be alkylated as shown in the following equation:
\[\ce{C6H5CH2NH2 + R - X -> C6H5CH2NHR}\]
Which of the following alkylhalides is best suited for this reaction through SN1 mechanism?
\[\ce{CH3Br}\]
\[\ce{C6H5Br}\]
\[\ce{C6H5CH2Br}\]
\[\ce{C2H5Br}\]
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
\[\ce{H2 (excess)/Pt}\]
\[\ce{LiAlH4}\] in ether
\[\ce{Fe}\] and \[\ce{HCl}\]
\[\ce{Sn}\] and \[\ce{HCl}\]
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
Sodium amide, \[\ce{NaNH2}\]
Sodium azide, \[\ce{NaN3}\]
Potassium cyanide, \[\ce{KCN}\]
Potassium phthalimide, \[\ce{C6H4 (CO)2N– K+}\]
The source of nitrogen in Gabriel synthesis of amines is ______.
Sodium azide, \[\ce{NaN3}\]
Sodium nitrite, \[\ce{NaNO2}\]
Potassium cyanide, \[\ce{KCN}\]
Potassium phthalimide, \[\ce{C6H4(CO)2N– K+}\]
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
\[\ce{2° R - Br + NH3}\]
\[\ce{2° R - Br + NaCN followed by H2 /Pt}\]
\[\ce{1° R - NH2 + RCHO followed by H2/Pt}\]
\[\ce{1° R - Br (2 mol) + potassium phthalimide followed by H3O+/heat}\]
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanamine is ______.
excess \[\ce{H2}\]
\[\ce{Br2}\] in aqueous \[\ce{NaOH}\]
iodine in the presence of red phosphorus
\[\ce{LiAlH4}\] in ether
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
\[\ce{excess H2/Pt}\]
\[\ce{NaOH/Br2}\]
\[\ce{NaBH4 /methanol}\]
\[\ce{LiAlH4 /ether}\]
Hoffmann Bromamide Degradation reaction is shown by ______.
\[\ce{ArNH2}\]
\[\ce{ArCONH2}\]
\[\ce{ArNO2}\]
\[\ce{ArCH2NH2}\]
The correct increasing order of basic strength for the following compounds is ______.
(I)

(II)

(III)

II < III < I
III < I < II
III < II < I
II < I < III
Methylamine reacts with \[\ce{HNO2}\] to form ______.
\[\ce{CH3 - O - N = O}\]
\[\ce{CH3 - O - CH3}\]
\[\ce{CH3OH}\]
\[\ce{CH3CHO}\]
The gas evolved when methylamine reacts with nitrous acid is:
\[\ce{NH3}\]
\[\ce{N2}\]
\[\ce{H2}\]
\[\ce{C2H6}\]
In the nitration of benzene using a mixture of conc. \[\ce{H2SO4}\] and conc. \[\ce{HNO3}\], the species which initiates the reaction is ______.
\[\ce{NO2}\]
\[\ce{NO+}\]
\[\ce{NO^{+}2}\]
\[\ce{NO^{-}2}\]
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
aromatic oxime
aromatic hydrocarbon
aromatic primary amine
aromatic amide
The most reactive amine towards dilute hydrochloric acid is:
CH3 – NH2
\[\begin{array}{cc}
\ce{H3C}\phantom{.............}\\\
\backslash\phantom{.......}\\
\ce{NH}\\
/\phantom{.......}\\
\ce{H3C}\phantom{...............}
\end{array}\]\[\begin{array}{cc}
\ce{H3C}\phantom{..........................}\\
\backslash\phantom{..................}\\
\ce{NH - CH3}\phantom{}\\
/\phantom{...................}\\
\ce{H3C}\phantom{...........................}\\
\end{array}\]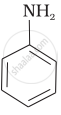
Acid anhydrides on reaction with primary amines give ______.
amide
imide
secondary amine
imine
The reaction \[\ce{Ar\overset{+}{N2}Cl- ->[Cu/HCl] ArCl + N2 + CuCl}\] is anmed as ______.
Sandmeyer reaction
Gatterman reaction
Claisen reaction
Carbylamine reaction
Best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is ______.
Hoffmann Bromamide reaction
Gabriel phthalimide synthesis
Sandmeyer reaction
Reaction with \[\ce{NH3}\]
Which of the following compound will not undergo azo coupling reaction with benzene diazonium chloride?
Aniline
Phenol
Anisole
Nitrobenzene
Which of the following compounds is the weakest Brönsted base?
Among the following amines, the strongest Brönsted base is:
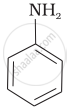
\[\ce{NH3}\]
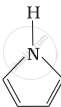
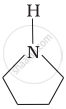
The correct decreasing order of basic strength of the following species is ______.
\[\ce{H2O, NH3, OH-, NH^{-}2}\]
\[\ce{NH^{-}2 > OH- > NH3 > H2O}\]
\[\ce{OH- > NH^{-}2 > H2O > NH3}\]
\[\ce{NH3 > H2O > NH^{-}2 > OH^-}\]
\[\ce{H2O > NH3 > OH- > NH^{-}2}\]
Which of the following should be most volatile?
\[\ce{(CH3)3N}\]
\[\ce{CH3CH2CH3}\]
\[\ce{CH3CH2CH2NH2}\]
\[\begin{array}{cc}
\ce{CH3CH2}\\
\phantom{..........}\backslash\\
\phantom{...............}\ce{NH}\\
\phantom{...........}/\\\
\phantom{......}\ce{CH3}
\end{array}\]
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Reaction of nitrite with \[\ce{LiAlH4}\].
Reaction of amide with \[\ce{LiAlH4}\] followed by treatment with water
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis.
Treatment of amide with bromine in aqueous solution of sodium hydroxide.
Which of the following cannot be prepared by Sandmeyer’s reaction?
(i) Chlorobenzene
(ii) Bromobenzene
(iii) Iodobenzene
(iv) Fluorobenzene
Reduction of nitrobenzene by which of the following reagent gives aniline?
(i) \[\ce{Sn/HCl}\]
(ii) \[\ce{Fe/HCl}\]
(iii) \[\ce{H2 - Pd}\]
(iv) \[\ce{Sn/NH4OH}\]
Which of the following species are involved in the carbylamine test?
(i) \[\ce{R - NC}\]
(ii) \[\ce{CHCl3}\]
(iii) \[\ce{COCl2}\]
(iv) \[\ce{NaNO2 + HCl}\]
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
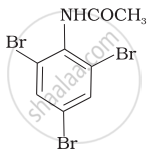
The product of the following reaction is:

(i)
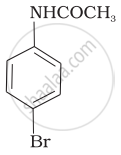
(ii)
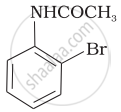
(iii)
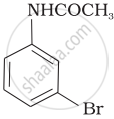
(iv)
Arenium ion involved in the bromination of aniline is:
(i)

(ii)

(iii)

(iv)

Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methylbenzylamine
(iv) Aniline
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(i) Acetyl chloride/pyridine followed by reaction with conc.\[\ce{H2SO4 }\] + conc. \[\ce{HNO3}\].
(ii) Acetic anyhdride/pyridine followed by conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iii) Dil. HCl followed by reaction with conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iv) Reaction with conc.\[\ce{HNO3}\] + conc.\[\ce{H2 SO4}\].
Which of the following reactions belong to electrophilic aromatic substitution?
(i) Bromination of acetanilide
(ii) Coupling reaction of aryldiazonium salts
(iii) Diazotisation of aniline
(iv) Acylation of aniline
What is the role of \[\ce{HNO3}\] in the nitrating mixture used for nitration of benzene?
Why is \[\ce{NH2}\] group of aniline acetylated before carrying out nitration?
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
What is the best reagent to convert nitrile to primary amine?
Give the structure of ‘A’ in the following reaction.

What is Hinsberg's reagent?
Why is benzene diazonium chloride not stored and is used immediately after its preparation?
Why does acetylation of –NH2 group of aniline reduce its activating effect?
Explain why \[\ce{MeNH2}\] is stronger base than \[\ce{MeOH}\]?
What is the role of pyridine in the acylation reaction of amines?
Under what reaction conditions (acidic/basic), the coupling reaction of aryldiazonium chloride with aniline is carried out?
Predict the product of reaction of aniline with bromine in non-polar solvent such as \[\ce{CS2}\].
Arrange the following compounds in increasing order of dipole moment.
\[\ce{CH3CH2CH3, CH3CH2NH2, CH3CH2OH}\]
What is the structure and IUPAC name of the compound, allylamine?
Write down the IUPAC name of 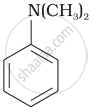
A compound Z with molecular formula \[\ce{C3H9N}\] reacts with \[\ce{C6H5SO2Cl}\] to give a solid, insoluble in alkali. Identify Z.
A primary amine, \[\ce{RNH2}\] can be reacted with \[\ce{CH3-X}\] to get secondary amine, \[\ce{R-NHCH3}\] but the only disadvantage is that 3° amine and quaternary ammonium salts are also obtained as side products. Can you suggest a method where \[\ce{RNH2}\] forms only 2° amine?
Complete the following reaction.
Why is aniline soluble in aqueous HCl?
Suggest a route by which the following conversion can be accomplished.

Identify A and B in the following reaction.

How will you carry out the following conversions?
toluene `->` p-toluidine
How will you carry out the following conversions?
p-toluidine diazonium chloride `→` p-toluic acid
Write following conversions:
nitrobenzene `->` acetanilide
Write following conversions:
acetanilide `->` p-nitroaniline
A solution contains 1 g mol. each of p-toluene diazonium chloride and p-nitrophenyl diazonium chloride. To this 1 g mol. of alkaline solution of phenol is added. Predict the major product. Explain your answer.
How will you bring out the following conversion?

How will you carry out the following conversion?

How will you carry out the following conversion?

How will you carry out the following conversions?

How will you carry out the following conversions?

Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Match the compounds given in Column I with the items given in Column II.
| Column I | Column II | ||
| (i) | Benzene sulphonyl chloride | (a) | Zwitter ion |
| (ii) | Sulphanilic acid | (b) | Hinsberg reagent |
| (iii) | Alkyl diazonium salts | (c) | Dyes |
| (iv) | Aryl diazonium salts | (d) | Conversion to alcohols |
Assertion: Acylation of amines gives a monosubstituted product whereas alkylation of amines gives polysubstituted product.
Reason: Acyl group sterically hinders the approach of further acyl groups.
Both assertion and reason are wrong.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Assertion is correct statement but reason is wrong statement.
Both assertion and reason are correct statements and reason is correct explanation of assertion.
Assertion is wrong statement but reason is correct statement.
Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
Both assertion and reason are wrong.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Assertion is correct statement but reason is wrong statement.
Both assertion and reason are correct statements and reason is correct explanation of assertion.
Assertion is wrong statement but reason is correct statement.
Assertion: N-Ethylbenzene sulphonamide is soluble in alkali.
Reason: Hydrogen attached to nitrogen in sulphonamide is strongly acidic.
Both assertion and reason are wrong.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Assertion is correct statement but reason is wrong statement.
Both assertion and reason are correct statements and reason is correct explanation of assertion.
Assertion is wrong statement but reason is correct statement.
Assertion: N, N-Diethylbenzene sulphonamide is insoluble in alkali.
Reason: Sulphonyl group attached to nitrogen atom is strong electron-withdrawing group.
Both assertion and reason are wrong.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Assertion is correct statement but reason is wrong statement.
Both assertion and reason are correct statements and reason is correct explanation of assertion.
Assertion is wrong statement but reason is correct statement.
Assertion: Only a small amount of \[\ce{HCl}\] is required in the reduction of nitro compounds with iron scrap and \[\ce{HCl}\] in the presence of steam.
Reason: \[\ce{FeCl2}\] formed gets hydrolysed to release \[\ce{HCl}\] during the reaction.
Both assertion and reason are wrong.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Assertion is correct statement but reason is wrong statement.
Both assertion and reason are correct statements and reason is correct explanation of assertion.
Assertion is wrong statement but reason is correct statement.
Assertion: Aromatic 1° amines can be prepared by Gabriel Phthalimide Synthesis.
Reason: Aryl halides undergo nucleophilic substitution with anion formed by phthalimide.
Both assertion and reason are wrong.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Assertion is correct statement but reason is wrong statement.
Both assertion and reason are correct statements and reason is correct explanation of assertion.
Assertion is wrong statement but reason is correct statement.
Assertion: Acetanilide is less basic than aniline.
Reason: Acetylation of aniline results in decrease of electron density on nitrogen.
Both assertion and reason are wrong.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Assertion is correct statement but reason is wrong statement.
Both assertion and reason are correct statements and reason is correct explanation of assertion.
Assertion is wrong statement but reason is correct statement.
A hydrocarbon ‘A’, \[\ce{(C4H8)}\] on reaction with \[\ce{HCl}\] gives a compound ‘B’, \[\ce{(C4H9Cl)}\], which on reaction with 1 mol of \[\ce{NH3}\] gives compound ‘C’, \[\ce{(C4H11N)}\]. On reacting with \[\ce{NaNO2}\] and \[\ce{HCl}\] followed by treatment with water, compound ‘C’ yields an optically active alcohol, ‘D’. Ozonolysis of ‘A’ gives 2 mols of acetaldehyde. Identify compounds ‘A’ to ‘D’. Explain the reactions involved.
A colourless substance ‘A’ \[\ce{(C6H7N)}\] is sparingly soluble in water and gives a water soluble compound ‘B’ on treating with mineral acid. On reacting with \[\ce{CHCl3}\] and alcoholic potash ‘A’ produces an obnoxious smell due to the formation of compound ‘C’. Reaction of ‘A’ with benzenesulphonyl chloride gives compound ‘D’ which is soluble in alkali. With \[\ce{NaNO2}\] and \[\ce{HCl}\], ‘A’ forms compound ‘E’ which reacts with phenol in alkaline medium to give an orange dye ‘F’. Identify compounds ‘A’ to ‘F’.
Predict the reagent or the product in the following reaction sequence.
Solutions for 13: Amines
![NCERT Exemplar solutions for Chemistry Exemplar [English] Class 12 chapter 13 - Amines NCERT Exemplar solutions for Chemistry Exemplar [English] Class 12 chapter 13 - Amines - Shaalaa.com](/images/chemistry-exemplar-english-class-12_6:ff5a05b4361741f5816a7388844a6202.png)
NCERT Exemplar solutions for Chemistry Exemplar [English] Class 12 chapter 13 - Amines
Shaalaa.com has the CBSE, Karnataka Board PUC Mathematics Chemistry Exemplar [English] Class 12 CBSE, Karnataka Board PUC solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. NCERT Exemplar solutions for Mathematics Chemistry Exemplar [English] Class 12 CBSE, Karnataka Board PUC 13 (Amines) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. NCERT Exemplar textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Chemistry Exemplar [English] Class 12 chapter 13 Amines are Concept of Amines, Classification of Amines, Structure of Amines, Physical Properties of Amines, Nomenclature of Animes, Uses of Amines, Identification of Primary, Secondary and Tertiary Amines, Cyanides and Isocyanides, Method of Preparation of Diazonium Salts, Diazonium Salts, Importance of Diazonium Salts in Synthesis of Aromatic Compounds, Preparation of Amines, Chemical Reactions of Amines - Basic Character of Amines, Chemical Reactions of Amines - Alkylation and Acylation, Chemical Reactions of Amines - Carbylamine Reaction, Chemical Reactions of Amines - Reaction with Nitrous Acid, Chemical Reactions of Amines - Reaction with Arylsulphonyl Chloride, Chemical Reactions of Amines - Electrophilic Substitution, Physical Properties of Diazonium Salts, Chemical Reaction of Diazonium Salts - Reactions Involving Displacement of Nitrogen, Chemical Reaction of Diazonium Salts - Reactions Involving Retention of Diazo Group, Organic Compounds Containing Nitrogen Numericals, Overview of Amines.
Using NCERT Exemplar Chemistry Exemplar [English] Class 12 solutions Amines exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in NCERT Exemplar Solutions are essential questions that can be asked in the final exam. Maximum CBSE, Karnataka Board PUC Chemistry Exemplar [English] Class 12 students prefer NCERT Exemplar Textbook Solutions to score more in exams.
Get the free view of Chapter 13, Amines Chemistry Exemplar [English] Class 12 additional questions for Mathematics Chemistry Exemplar [English] Class 12 CBSE, Karnataka Board PUC, and you can use Shaalaa.com to keep it handy for your exam preparation.