Advertisements
Advertisements
Question
Hoffmann Bromamide Degradation reaction is shown by ______.
Options
\[\ce{ArNH2}\]
\[\ce{ArCONH2}\]
\[\ce{ArNO2}\]
\[\ce{ArCH2NH2}\]
Advertisements
Solution
Hoffmann Bromamide Degradation reaction is shown by \[\ce{ArCONH2}\].
Explanation:
Hoffmann invented a method for producing primary amines by treating an amide with bromine in an aqueous or ethanolic sodium hydroxide solution. An alkyl or aryl group migrates from the amide's carbonyl carbon to the nitrogen atom during this degradation reaction.
\[\ce{C4H5CONH2 ->[Br2 + NaOH] C4H5NH2}\]
APPEARS IN
RELATED QUESTIONS
Give the structures of A, B and C in the following reactions :

Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Write structures of compounds A and B in each of the following reactions:
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
Identify the product obtained when benzyl chloride undergoes ammonolysis in presence of excess ammonia followed by the reaction with two moles of methyl iodide.
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
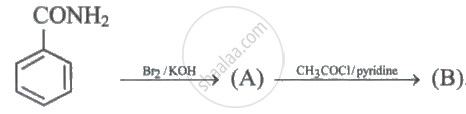
Identify the major product (B).
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which of the following reagents is used in Hofmann's elimination reaction of amines?
Which of the following does NOT give carbylamine test?
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
Among the following amines, the strongest Brönsted base is:
Reduction of nitrobenzene by which of the following reagent gives aniline?
(i) \[\ce{Sn/HCl}\]
(ii) \[\ce{Fe/HCl}\]
(iii) \[\ce{H2 - Pd}\]
(iv) \[\ce{Sn/NH4OH}\]
Write following conversions:
nitrobenzene `->` acetanilide
How will you carry out the following conversion?

Ethylamine can be prepared by the action of bromine and caustic potash on which compound?
Which of the following compound gives pink colour on reaction with phthalic anhydride in cone. H2SO4 followed by treatment with NaOH?
Write a short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis.
Write short notes on the following:
Ammonolysis
