Advertisements
Advertisements
Question
Write following conversions:
nitrobenzene `->` acetanilide
Advertisements
Solution
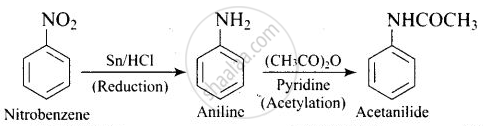
APPEARS IN
RELATED QUESTIONS
Accomplish the following conversion:
Benzamide to toluene
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2Br ->[KCN] A ->[LiAlH4] B ->[HNO2][0^\circ C] C}\]
Mention 'two' uses of propan-2-one.
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Account for the following:
Gabriel phthalimide synthesis is not preferred for preparing aromatic primary amines.
Write reactions to prepare ethanamine from Acetonitrile.
Acetamide on reduction using Na/C2H5OH gives ____________.
Identify the product 'A' in the following reaction.
\[\ce{Aniline ->[(CH3CO)2O][Pyridine] A}\]
Alkyl cyanides on reduction by sodium and ethanol give primary amines. This reaction is called as ____________.
Identify the product obtained when benzyl chloride undergoes ammonolysis in presence of excess ammonia followed by the reaction with two moles of methyl iodide.
____________ can be prepared exclusively by Gabriel phthalimide synthesis.
What product is formed when \[\ce{R - C ≡ N}\] is hydrolysed?
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds:
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanamine is ______.
Assertion: Aromatic 1° amines can be prepared by Gabriel Phthalimide Synthesis.
Reason: Aryl halides undergo nucleophilic substitution with anion formed by phthalimide.
Account for the following:
Aniline cannot be prepared by the ammonolysis of chlorobenzene under normal conditions.
The amine 'A' when treated with nitrous acid gives yellow oily substance. The amine A is ______.
Which of the following statement(s) is/are incorrect in case of Hofmann bromamide degradation?
Write a short note on the following:
Ammonolysis
Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
