Advertisements
Advertisements
Question
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanamine is ______.
Options
excess \[\ce{H2}\]
\[\ce{Br2}\] in aqueous \[\ce{NaOH}\]
iodine in the presence of red phosphorus
\[\ce{LiAlH4}\] in ether
Advertisements
Solution
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanamine is \[\ce{LiAlH4}\] in ether.
Explanation:

APPEARS IN
RELATED QUESTIONS
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Accomplish the following conversion:
Benzyl chloride to 2-phenylethanamine
Give the structures of A, B and C in the following reactions :
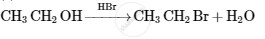
Give the structures of A, B and C in the following reactions :

Name the process of breaking C-X bond by ammonia in preparation of amines.
Write the order of reactivity of alkyl halides with ammonia.
In aqueous phase the order of basic strength of alkylamine is ______.
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Reduction of nitrobenzene by which of the following reagent gives aniline?
(i) \[\ce{Sn/HCl}\]
(ii) \[\ce{Fe/HCl}\]
(iii) \[\ce{H2 - Pd}\]
(iv) \[\ce{Sn/NH4OH}\]
Identify A and B in the following reaction.

Write following conversions:
acetanilide `->` p-nitroaniline
How will you carry out the following conversions?

Give reasons for the following:
Ammonolysis of alkyl halides is not a good method to prepare pure primary amines.
Which of the following compound gives pink colour on reaction with phthalic anhydride in cone. H2SO4 followed by treatment with NaOH?
Write the name of the product formed by the action of LiAlH4/ether on acetamide.
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Write short note on the following:
Ammonolysis
Assertion: Amimonolysis of alkyl halides involves the reaction between alkyl halides and alcoholic ammonia.
Reason: Ammonolysis of alkyl halides produces secondary amines only.
Write a short note on the following:
Ammonolysis.
