Advertisements
Advertisements
Question
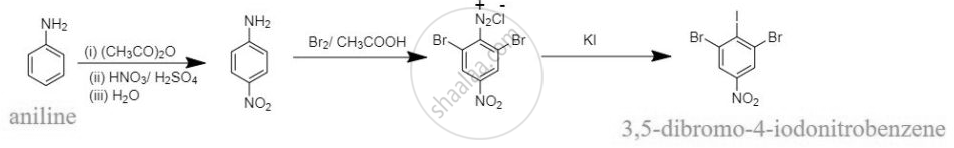
How will you carry out the following conversions?

Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
How are propan-1-amine and propan-2-amine prepared from oxime?
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Give the structures of A, B and C in the following reactions :

Answer in one sentence.
Which amide does produce ethanamine by Hofmann bromamide degradation reaction?
Answer the following
Explain the ammonolysis of alkyl halides.
Write the order of reactivity of alkyl halides with ammonia.
Alkyl cyanides on reduction by sodium and ethanol give primary amines. This reaction is called as ____________.
____________ can be prepared exclusively by Gabriel phthalimide synthesis.
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(i) Acetyl chloride/pyridine followed by reaction with conc.\[\ce{H2SO4 }\] + conc. \[\ce{HNO3}\].
(ii) Acetic anyhdride/pyridine followed by conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iii) Dil. HCl followed by reaction with conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iv) Reaction with conc.\[\ce{HNO3}\] + conc.\[\ce{H2 SO4}\].
Suggest a route by which the following conversion can be accomplished.

Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
Account for the following:
Aniline cannot be prepared by the ammonolysis of chlorobenzene under normal conditions.
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
When primary amines are treated with HCl, the product obtained is which of the following?
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Write a short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis.
