Advertisements
Advertisements
Question
Write the order of reactivity of alkyl halides with ammonia.
Advertisements
Solution
The order of reactivity of alkyl halides with ammonia is, R–I > R–Br > R–Cl.
RELATED QUESTIONS
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

How do you convert the following: Ethanenitrile to ethanamine
Give the structures of A, B and C in the following reactions :

Give the structure of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2 + HCl][273 K] B ->[H2O/H+][\Delta] C}\]
Give a plausible explanation for the following:
Why are amines less acidic than alcohols of comparable molecular masses?
Give the structures of A, B and C in the following reactions :
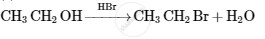
Choose the most correct option.
Which of the following compounds will dissolve in aqueous NaOH after undergoing reaction with Hinsberg reagent?
Answer in one sentence.
Predict the product of the following reaction.
\[\ce{Nitrobenzene ->[Sn/conc.HCl]?}\]
Answer the following
Write a reaction to convert acetic acid into methylamine.
Answer the following
Explain Gabriel phthalimide synthesis.
Mendius reaction is used to convert _____________
Name the process of breaking C-X bond by ammonia in preparation of amines.
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Write reactions for the preparation of ethanamine using Gabriel phthalimide synthesis.
Why cannot aniline be prepared by Gabriel phthalimide synthesis?
Explain the following reaction with a suitable example.
Hofmann elimination reaction
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
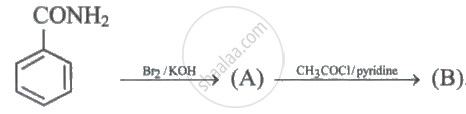
Identify the major product (B).
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
Which of the following reactions is appropriate for converting benzamide to aniline?
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Which of the following reagents is used in Hofmann's elimination reaction of amines?
Which of the following does NOT give carbylamine test?
Which of the following reactions does NOT yield an amine?
For producing amines, the reaction of nitro compounds with iron scrap is preferred because:
Amongst the following, the strongest base in aqueous medium is ______.
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
Hoffmann Bromamide Degradation reaction is shown by ______.
Which of the following compounds is the weakest Brönsted base?
Reduction of nitrobenzene by which of the following reagent gives aniline?
(i) \[\ce{Sn/HCl}\]
(ii) \[\ce{Fe/HCl}\]
(iii) \[\ce{H2 - Pd}\]
(iv) \[\ce{Sn/NH4OH}\]
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

What is the best reagent to convert nitrile to primary amine?
Identify A and B in the following reaction.

How will you carry out the following conversion?

How will you carry out the following conversions?

How will you carry out the following conversions?

Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
A primary amine is formed by an amide on treatment with bromine and alkali. The primary amine has
The compound X is which of the following?
\[\ce{CH3CN ->[Na + C2H5OH] x}\]
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Amides can be converted into amines by the reaction named ______.
Write short note on the following:
Ammonolysis
Write short note on the following:
Ammonolysis
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
Write a short note on the following:
Ammonolysis
Assertion: Amimonolysis of alkyl halides involves the reaction between alkyl halides and alcoholic ammonia.
Reason: Ammonolysis of alkyl halides produces secondary amines only.
Write a short note on the following:
Ammonolysis.
