Advertisements
Advertisements
Question
Give a plausible explanation for the following:
Why are amines less acidic than alcohols of comparable molecular masses?
Advertisements
Solution
Loss of a proton from an amine gives an amide ion, while loss of a proton from alcohol gives an alkoxide ion.
\[\ce{R-NH2 -> R-NH– + H+}\]
\[\ce{R-O-H -> R-O^- + H+}\]
Since O is more electronegative than N, it will attract positive species more strongly in comparison to N. Thus, RO− is more stable than RNH−. Thus, alcohols are more acidic than amines. Conversely, amines are less acidic than alcohols.
APPEARS IN
RELATED QUESTIONS
How are propan-1-amine and propan-2-amine prepared from oxime?
Accomplish the following conversion:
Benzamide to toluene
Give the structures of A, B and C in the following reactions :
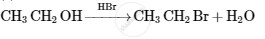
Why cannot aniline be prepared by Gabriel phthalimide synthesis?
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Which of the following compounds is obtained when quaternary ammonium hydroxide is strongly heated?
Which of the following reactions does NOT yield an amine?
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
What is the best reagent to convert nitrile to primary amine?
Identify A and B in the following reaction.

Write following conversions:
nitrobenzene `->` acetanilide
How will you carry out the following conversion?

How will you carry out the following conversions?

A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
Amides can be converted into amines by the reaction named ______.
Write a short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis.
