Advertisements
Advertisements
Question
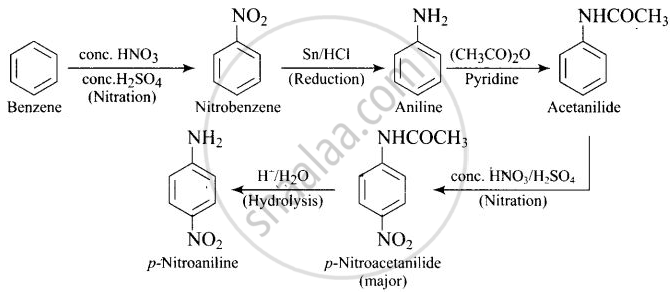
How will you carry out the following conversion?

Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
Identify 'A' and 'B' in the following reaction and rewrite the complete reaction :

Give the structures of A, B and C in the following reactions :

Answer in one sentence.
Which amide does produce ethanamine by Hofmann bromamide degradation reaction?
Answer the following
Identify A and B in the following reactions.
\[\ce{C6H5CH2Br->[alco.][KCN]A ->[Na/ethanol]B.}\]
The following amines is the product of Gabriel phthalimide synthesis.
Name the process of breaking C-X bond by ammonia in preparation of amines.
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Which of the following reactions is appropriate for converting benzamide to aniline?
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
Which of the following compounds is obtained when quaternary ammonium hydroxide is strongly heated?
Among the following amines, the strongest Brönsted base is:
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
The compound X is which of the following?
\[\ce{CH3CN ->[Na + C2H5OH] x}\]
Reduction of nitro alkanes yields which compound?
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
The amine 'A' when treated with nitrous acid gives yellow oily substance. The amine A is ______.
Write short note on the following:
Ammonolysis
Identify the compo ds A and B in the following reactions:
\[\ce{A ->[Nitrating mixture] B ->[(i) Sn/cone. HCI][(ii) NaOH] Aniline}\]
Write a short note on the following:
Ammonolysis
