Advertisements
Advertisements
प्रश्न
How will you carry out the following conversion?

Advertisements
उत्तर
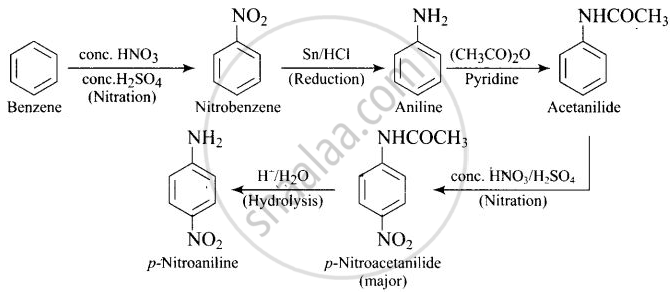
APPEARS IN
संबंधित प्रश्न
Give the structures of A, B and C in the following reactions :

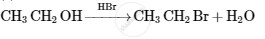
Accomplish the following conversion:
Benzyl chloride to 2-phenylethanamine
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2 + HCl][273 K] B ->[H2O/H+][\Delta] C}\]
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Give the structures of A, B and C in the following reactions :
The following amines is the product of Gabriel phthalimide synthesis.
Write reactions for the preparation of ethanamine using Gabriel phthalimide synthesis.
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
Identify the product obtained, when benzamide is treated with bromine and aqueous sodium hydroxide.
What product is formed when \[\ce{R - C ≡ N}\] is hydrolysed?
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
Best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is ______.
How will you bring out the following conversion?

Which of the following statement(s) is/are incorrect in case of Hofmann bromamide degradation?
Identify the product ‘C’ in the following reaction.
\[\ce{Aniline ->[(CH3CH)2O][Pyridine] A ->[Br2][CH3COOH] B ->[H^+ or OH^-] C}\]
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Write short notes on the following:
Ammonolysis
