Advertisements
Advertisements
प्रश्न
Give the structures of A, B and C in the following reactions :
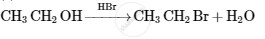
Advertisements
उत्तर
The reaction proceeds through nucleophilic substitution bimolecular (SN2) mechanism, as shown:

APPEARS IN
संबंधित प्रश्न
How do you convert the following: Ethanenitrile to ethanamine
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2Br ->[KCN] A ->[LiAlH4] B ->[HNO2][0^\circ C] C}\]
Write the reaction of aliphatic primary amine with nitrous acid.
Explain the following reaction with a suitable example.
Hofmann elimination reaction
Identify 'A' and 'B' in the following conversions.
\[\ce{CH3 - I ->[Alc. KCN][\Delta] A ->[Na/C2H5OH] B}\]
Quaternary ammonium salt is formed:
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
Identify the compo ds A and B in the following reactions:
\[\ce{A ->[Nitrating mixture] B ->[(i) Sn/cone. HCI][(ii) NaOH] Aniline}\]
Write short note on the following:
Ammonolysis
