Advertisements
Advertisements
प्रश्न
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
विकल्प
LiAlH4
H2/Ni
Fe and HCl
Sn and HCl
Advertisements
उत्तर
LiAlH4
Explanation:
When nitrobenzene interacts with LiAlH4 in an ether medium, it does not change into amine; instead, azobenzene is produced. As a result, this reagent is not suitable for converting nitrobenzene to amine.
APPEARS IN
संबंधित प्रश्न
Write a short note on Hoffmann bromamide degradation.
Mention 'two' uses of propan-2-one.
Write reactions to bring about the following conversions.
Acetamide to methylamine
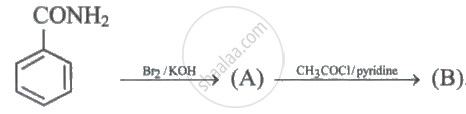
Identify the major product (B).
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Which of the following amines cannot be prepared by Gabriel phthalimide synthesis?
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
The compound X is which of the following?
\[\ce{CH3CN ->[Na + C2H5OH] x}\]
The amine 'A' when treated with nitrous acid gives yellow oily substance. The amine A is ______.
Write a short note on the following:
Ammonolysis
